Mechanism of constitutive export from the golgi: bulk flow via the formation, protrusion, and en bloc cleavage of large trans-golgi network tubular domains
- PMID: 12937271
- PMCID: PMC266766
- DOI: 10.1091/mbc.e03-01-0033
Mechanism of constitutive export from the golgi: bulk flow via the formation, protrusion, and en bloc cleavage of large trans-golgi network tubular domains
Abstract
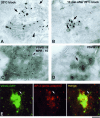
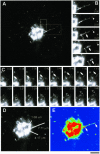
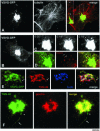
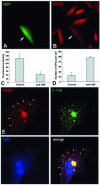
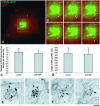
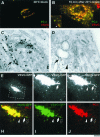
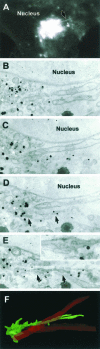
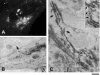
Transport of constitutive cargo proteins from the Golgi complex to the plasma membrane (PM) is known to be mediated by large tubular-saccular carriers moving along microtubules. However, the process by which these large structures emerge from the trans-Golgi network (TGN) remains unclear. Here, we address the question of the formation of Golgi-to-PM carriers (GPCs) by using a suitable cluster of morphological techniques, providing an integrated view of their dynamics and three-dimensional structure. Our results indicate that exit from the TGN of a constitutive traffic marker, the VSVG protein, occurs by bulk flow and is a three-step process. First, the formation of a tubular-reticular TGN domain (GPC precursor) that includes PM-directed proteins and excludes other cargo and Golgi-resident proteins. Notably, this step does not require membrane fusion. Second, the docking of this preformed domain on microtubules and its kinesin-mediated extrusion. Finally, the detachment of the extruded domain by membrane fission. The formation of GPCs does not involve cargo concentration and is not associated with the presence of known coat proteins on GPC precursors. In summary, export from the Golgi occurs via the formation, protrusion and en bloc cleavage of specialized TGN tubular-saccular domains.
Figures

References
-
- Acharya, U., Jacobs, R., Peters, J.M., Watson, N., Farquhar, M.G., and Malhotra, V. (1995). The formation of Golgi stacks from vesiculated Golgi membranes requires two distinct fusion events. Cell 82, 895-904. - PubMed
-
- Band, A.M., Maatta, J., Kaariainen, L., and Kuismanen, E. (2001). Inhibition of the membrane fusion machinery prevents exit from the TGN and proteolytic processing by furin. FEBS Lett. 505, 118-124. - PubMed
-
- Banting, G., and Ponnambalam, S. (1997). TGN38 and its orthologues: roles in post-TGN vesicle formation and maintenance of TGN morphology. Biochim. Biophys. Acta 1355, 209-217. - PubMed
-
- Brown, W.J., Chambers, K., and Doody, A. (2003). Phospholipase A2 (PLA2) enzymes in membrane trafficking: mediators of membrane shape and function. Traffic 4, 214-221. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

