Contribution of Ca2+-dependent conductances to membrane potential fluctuations of medullary respiratory neurons of newborn rats in vitro
- PMID: 12937288
- PMCID: PMC2343467
- DOI: 10.1113/jphysiol.2003.049312
Contribution of Ca2+-dependent conductances to membrane potential fluctuations of medullary respiratory neurons of newborn rats in vitro
Abstract
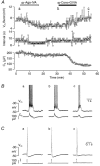
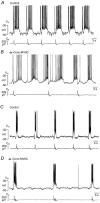
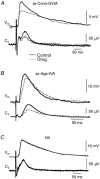
Ca2+-dependent conductances were studied in respiratory interneurons in the brainstem-spinal cord preparation of newborn rats. omega-Conotoxin-GVIA attenuated evoked postsynaptic potentials, spontaneous or evoked inspiratory spinal nerve activity and blocked spike afterhyperpolarization. Furthermore, omega-conotoxin-GVIA augmented rhythmic drive potentials of pre-inspiratory and inspiratory neurons and increased respiratory-related spike frequency of pre-inspiratory cells with no effect on inspiratory hyperpolarization. In contrast, omega-agatoxin-IVA depressed drive potentials of pre-inspiratory and inspiratory neurons and attenuated inspiratory hyperpolarization and spike frequency of pre-inspiratory cells. It did not affect spike shape and exerted only minor, non-significant, attenuating effects on spontaneous or evoked nerve bursts or evoked postsynaptic potentials. Nifedipine diminished drive potentials and spike frequency of pre-inspiratory neurons and shortened drive potentials in some cells. omega-Conotoxin-MVIIC attenuated drive potentials and intraburst firing rate of pre-inspiratory neurons and decreased substantially respiratory frequency. Respiratory rhythm disappeared following combined application of omega-conotoxin-GVIA, omega-conotoxin-MVIIC, omega-agatoxin-IVA and nifedipine. Apamin potentiated drive potentials and abolished spike afterhyperpolarization, whereas charybdotoxin and tetraethylammonium prolonged spike duration without effect on shape of drive potentials. The results show that specific sets of voltage-activated L-, N- and P/Q-type Ca2+ channels determine the activity of particular subclasses of neonatal respiratory neurons, whereas SK- and BK-type K+ channels attenuate drive potentials and shorten spikes, respectively, independent of cell type. We hypothesize that modulation of spontaneous activity of pre-inspiratory neurons via N-, L- and P/Q-type Ca2+ channels is important for respiratory rhythm or pattern generation.
Figures






Similar articles
-
Effects of various K+ channel blockers on spontaneous glycine release at rat spinal neurons.Brain Res. 2007 Jul 9;1157:11-22. doi: 10.1016/j.brainres.2006.09.097. Epub 2007 Jun 6. Brain Res. 2007. PMID: 17555723
-
Nifedipine- and omega-conotoxin-sensitive Ca2+ conductances in guinea-pig substantia nigra pars compacta neurones.J Physiol. 1993 Jul;466:727-47. J Physiol. 1993. PMID: 8410714 Free PMC article.
-
Calcium-dependent responses in neurons of the isolated respiratory network of newborn rats.J Physiol. 1996 Mar 15;491 ( Pt 3)(Pt 3):677-95. doi: 10.1113/jphysiol.1996.sp021249. J Physiol. 1996. PMID: 8815203 Free PMC article.
-
Neuronal mechanisms of respiratory rhythm generation: an approach using in vitro preparation.Jpn J Physiol. 1997 Oct;47(5):385-403. doi: 10.2170/jjphysiol.47.385. Jpn J Physiol. 1997. PMID: 9504127 Review.
-
Respiratory network function in the isolated brainstem-spinal cord of newborn rats.Prog Neurobiol. 1999 Dec;59(6):583-634. doi: 10.1016/s0301-0082(99)00009-x. Prog Neurobiol. 1999. PMID: 10845755 Review.
Cited by
-
Modulation of BK Channel Function by Auxiliary Beta and Gamma Subunits.Int Rev Neurobiol. 2016;128:51-90. doi: 10.1016/bs.irn.2016.03.015. Epub 2016 Apr 8. Int Rev Neurobiol. 2016. PMID: 27238261 Free PMC article. Review.
-
Metabotropic glutamate receptors activate dendritic calcium waves and TRPM channels which drive rhythmic respiratory patterns in mice.J Physiol. 2008 May 1;586(9):2277-91. doi: 10.1113/jphysiol.2007.149021. Epub 2008 Feb 28. J Physiol. 2008. PMID: 18308826 Free PMC article.
-
Cholinergic modulation of upper airway control: maturational changes and mechanisms at cellular and synaptic levels.J Neurophysiol. 2025 Jan 1;133(1):46-59. doi: 10.1152/jn.00165.2024. Epub 2024 Nov 28. J Neurophysiol. 2025. PMID: 39607299 Free PMC article. Review.
-
The respiratory control mechanisms in the brainstem and spinal cord: integrative views of the neuroanatomy and neurophysiology.J Physiol Sci. 2017 Jan;67(1):45-62. doi: 10.1007/s12576-016-0475-y. Epub 2016 Aug 17. J Physiol Sci. 2017. PMID: 27535569 Free PMC article. Review.
-
Mechanisms of CO2/H+ chemoreception by respiratory rhythm generator neurons in the medulla from newborn rats in vitro.J Physiol. 2006 Apr 15;572(Pt 2):525-37. doi: 10.1113/jphysiol.2005.102533. Epub 2006 Feb 9. J Physiol. 2006. PMID: 16469786 Free PMC article.
References
-
- Arata A, Onimaru H, Homma I. Respiration-related neurons in the ventral medulla of newborn rats in vitro. Brain Res Bull. 1990;24:599–604. - PubMed
-
- Ballanyi K, Onimaru H, Homma I. Respiratory network function in the isolated brainstem-spinal cord of newborn rats. Prog Neurobiol. 1999;59:583–634. - PubMed
-
- Bianchi AL, Denavit-Saubie M, Champagnat J. Central control of breathing in mammals: neuronal circuitry, membrane properties, and neurotransmitters. Physiol Rev. 1995;75:1–45. - PubMed
-
- Brockhaus J, Ballanyi K. Synaptic inhibition in the isolated respiratory network of neonatal rats. Eur J Neurosci. 1998;10:3823–3839. - PubMed
-
- Brockhaus J, Ballanyi K. Anticonvulsant A1 receptor-mediated adenosine action on neuronal networks in the brainstem-spinal cord of newborn rats. Neuroscience. 2000;96:359–371. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

