Interaction of Aurora-A and centrosomin at the microtubule-nucleating site in Drosophila and mammalian cells
- PMID: 12939255
- PMCID: PMC2172831
- DOI: 10.1083/jcb.200305048
Interaction of Aurora-A and centrosomin at the microtubule-nucleating site in Drosophila and mammalian cells
Abstract
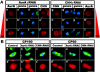
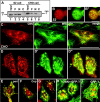
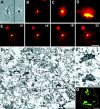
A mitosis-specific Aurora-A kinase has been implicated in microtubule organization and spindle assembly in diverse organisms. However, exactly how Aurora-A controls the microtubule nucleation onto centrosomes is unknown. Here, we show that Aurora-A specifically binds to the COOH-terminal domain of a Drosophila centrosomal protein, centrosomin (CNN), which has been shown to be important for assembly of mitotic spindles and spindle poles. Aurora-A and CNN are mutually dependent for localization at spindle poles, which is required for proper targeting of gamma-tubulin and other centrosomal components to the centrosome. The NH2-terminal half of CNN interacts with gamma-tubulin, and induces cytoplasmic foci that can initiate microtubule nucleation in vivo and in vitro in both Drosophila and mammalian cells. These results suggest that Aurora-A regulates centrosome assembly by controlling the CNN's ability to targeting and/or anchoring gamma-tubulin to the centrosome and organizing microtubule-nucleating sites via its interaction with the COOH-terminal sequence of CNN.
Figures

References
-
- Berdnik, D., and J.A. Knoblich. 2002. Drosophila Aurora-A is required for centrosome maturation and actin-dependent asymmetric protein localization during mitosis. Curr. Biol. 12:640–647. - PubMed
-
- Eyers, P.A., E. Erikson, L.G. Chen, and J.L. Maller. 2003. A novel mechanism for activation of the protein kinase Aurora A. Curr. Biol. 13:691–697. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

