Plasma membrane budding as an alternative release mechanism of the extracellular enveloped form of vaccinia virus from HeLa cells
- PMID: 12941903
- PMCID: PMC224582
- DOI: 10.1128/jvi.77.18.9931-9942.2003
Plasma membrane budding as an alternative release mechanism of the extracellular enveloped form of vaccinia virus from HeLa cells
Abstract
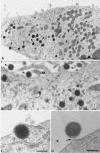
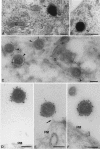
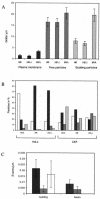
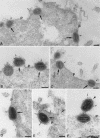
In HeLa cells the assembly of modified vaccinia virus Ankara (MVA), an attenuated vaccinia virus (VV) strain, is blocked. No intracellular mature viruses (IMVs) are made and instead, immature viruses accumulate, some of which undergo condensation and are released from the cell. The condensed particles may undergo wrapping by membranes of the trans-Golgi network and fusion with the plasma membrane prior to their release (M. W. Carroll and B. Moss, Virology 238:198-211, 1997). The present study shows by electron microscopy (EM), however, that the dense particles made in HeLa cells are also released by a budding process at the plasma membrane. By labeling the plasma membrane with antibodies to B5R, a membrane protein of the extracellular enveloped virus, we show that budding occurs at sites that concentrate this protein. EM quantitation revealed that the cell surface around a budding profile was as strongly labeled with anti-B5R antibody as were the extracellular particles, whereas the remainder of the plasma membrane was significantly less labeled. To test whether budding was a characteristic of MVA infection, HeLa cells were infected with the replication competent VV strains Western Reserve strain (WR) and International Health Department strain-J (IHD-J) and also prepared for EM. EM analyses, surprisingly, revealed for both virus strains IMVs that evidently budded at the cell surface at sites that were significantly labeled with anti-B5R. EM also indicated that budding of MVA dense particles was more efficient than budding of IMVs from WR- or IHD-J-infected cells. This was confirmed by semipurifying [(35)S]methionine-labeled dense particles or extracellular enveloped virus (EEVs) from the culture supernatant of MVA- or IHD-J-infected HeLa cells, respectively, showing that threefold more labeled dense particles were secreted than EEVs. Finally, although the released MVA dense particles contain some DNA, they are not infectious, as assessed by plaque assays.
Figures

Similar articles
-
Comparison of virus production in chicken embryo fibroblasts infected with the WR, IHD-J and MVA strains of vaccinia virus: IHD-J is most efficient in trans-Golgi network wrapping and extracellular enveloped virus release.J Gen Virol. 2003 Jun;84(Pt 6):1383-1392. doi: 10.1099/vir.0.19016-0. J Gen Virol. 2003. PMID: 12771405
-
The block in assembly of modified vaccinia virus Ankara in HeLa cells reveals new insights into vaccinia virus morphogenesis.J Virol. 2002 Aug;76(16):8318-34. doi: 10.1128/jvi.76.16.8318-8334.2002. J Virol. 2002. PMID: 12134037 Free PMC article.
-
Deletion of the vaccinia virus B5R gene encoding a 42-kilodalton membrane glycoprotein inhibits extracellular virus envelope formation and dissemination.J Virol. 1993 Aug;67(8):4732-41. doi: 10.1128/JVI.67.8.4732-4741.1993. J Virol. 1993. PMID: 8331727 Free PMC article.
-
The exit of vaccinia virus from infected cells.Virus Res. 2004 Dec;106(2):189-97. doi: 10.1016/j.virusres.2004.08.015. Virus Res. 2004. PMID: 15567497 Review.
-
Assembly of vaccinia virus revisited: de novo membrane synthesis or acquisition from the host?Trends Microbiol. 2002 Jan;10(1):15-24. doi: 10.1016/s0966-842x(01)02256-9. Trends Microbiol. 2002. PMID: 11755081 Review.
Cited by
-
Orthopoxvirus targets for the development of new antiviral agents.Antiviral Res. 2012 May;94(2):111-25. doi: 10.1016/j.antiviral.2012.02.012. Epub 2012 Mar 8. Antiviral Res. 2012. PMID: 22406470 Free PMC article. Review.
-
Vaccinia virus entry into cells is dependent on a virion surface protein encoded by the A28L gene.J Virol. 2004 Mar;78(5):2357-66. doi: 10.1128/jvi.78.5.2357-2366.2004. J Virol. 2004. PMID: 14963132 Free PMC article.
-
Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution.Viruses. 2020 Nov 5;12(11):1257. doi: 10.3390/v12111257. Viruses. 2020. PMID: 33167496 Free PMC article. Review.
-
Conserved Oligomeric Golgi (COG) Complex Proteins Facilitate Orthopoxvirus Entry, Fusion and Spread.Viruses. 2020 Jun 30;12(7):707. doi: 10.3390/v12070707. Viruses. 2020. PMID: 32629851 Free PMC article.
-
Monkeypox Virus Host Factor Screen Using Haploid Cells Identifies Essential Role of GARP Complex in Extracellular Virus Formation.J Virol. 2017 May 12;91(11):e00011-17. doi: 10.1128/JVI.00011-17. Print 2017 Jun 1. J Virol. 2017. PMID: 28331092 Free PMC article.
References
-
- Antoine, G., F. Scheiflinger, F. Dorner, and F. G. Falkner. 1998. The complete genomic sequence of the modified vaccinia ankara strain: comparison with other orthopoxviruses. Virology 244:365-396. - PubMed
-
- Boulanger, D., T. Smith, and M. A. Skinner. 2000. Morphogenesis and release of fowlpox virus. J. Gen. Virol. 81:675-687. - PubMed
-
- Carroll, M. W., and B. Moss. 1997. Host range and cytopathogenicity of the highly attenuated MVA strain of vaccinia virus: propagation and generation of recombinant viruses in a nonhuman mammalian cell line. Virology 238:198-211. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

