Condensed chromatin domains in the mammalian nucleus are accessible to large macromolecules
- PMID: 12947417
- PMCID: PMC1326359
- DOI: 10.1038/sj.embor.embor922
Condensed chromatin domains in the mammalian nucleus are accessible to large macromolecules
Abstract
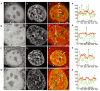
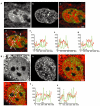
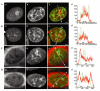
Most chromatin in interphase nuclei is part of condensed chromatin domains. Previous work has indicated that transcription takes place primarily at the surface of chromatin domains, that is, in the perichromatin region. It is possible that genes inside chromatin domains are silenced due to inaccessibility to macromolecular components of the transcription machinery. We have tested the accessibility of chromatin domains in nuclei of living cells with proteins and dextrans of different molecular sizes. Our results show that chromatin domains are readily accessible to large macromolecules, including proteins with a molecular weight of several hundred kilodaltons. Therefore, the silencing of genes that are incorporated into such domains is not due to the physical inaccessibility of condensed chromatin domains to transcription factors.
Figures
References
-
- Brown K.E., Baxter J., Graf D., Merkenschlager M. & Fisher A.G. ( 1999) Dynamic repositioning of genes in the nucleus of lymphocytes preparing for cell division. Mol. Cell, 3, 207–217. - PubMed
-
- Chubb J.R. & Bickmore W.A. ( 2003) Considering nuclear comparmentalization in the light of nuclear compartments. Cell, 112, 403–406. - PubMed
-
- Chubb J.R., Boyle S., Perry P. & Bickmore W.A. ( 2002) Chromatin motion is constrained by association with nuclear compartments in human cells. Curr. Biol., 12, 439–445. - PubMed
-
- Cockell M. & Gasser S.M. ( 1999) Nuclear compartments and gene regulation. Curr. Opin. Genet. Dev., 9, 199–205. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

