Calcium regulation of the soluble adenylyl cyclase expressed in mammalian spermatozoa
- PMID: 12958208
- PMCID: PMC196863
- DOI: 10.1073/pnas.1831008100
Calcium regulation of the soluble adenylyl cyclase expressed in mammalian spermatozoa
Abstract
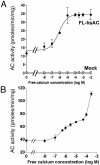
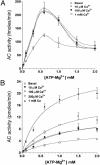
In mammals, Ca2+ and HCO3- ions play a critical role in the regulation of sperm function, most likely by regulation of cAMP levels. Mammalian germ cells contain a soluble adenylyl cyclase (sAC) with properties distinct from the well characterized membrane-bound enzymes Here we investigated whether the cyclase expressed in mature spermatozoa has the properties of sAC and whether it is regulated by Ca2+. In addition to an HCO3--dependent activation, the cyclase endogenous to human spermatozoa is stimulated 2- to 3-fold by Ca2+ in a concentration-dependent manner (EC50 approximately 400 nM). In a similar fashion, Ca2+ activates the recombinant rat and human full-length sAC with similar EC50 values. The Ca2+ stimulation was also observed when sAC was activated with HCO3-, was independent of calmodulin, and was associated with an increase in Vmax without changes in Km for ATP-Mg2+. An increase in intracellular Ca2+ by ionophore or by a muscarinic cholinergic receptor agonist increases cAMP in cells transfected with FL-hsAC, but not in mock-transfected cells. Similarly, both Ca2+ and HCO3- stimulate cAMP accumulation in human spermatozoa. These findings provide evidence that human spermatozoa express a cyclase with the properties of sAC and that Ca2+ can substitute for HCO3- in the stimulation of this enzyme, underscoring an important role for sAC in the control of sperm functions.
Figures

Similar articles
-
Identification and functional analysis of splice variants of the germ cell soluble adenylyl cyclase.J Biol Chem. 2001 Aug 24;276(34):31698-708. doi: 10.1074/jbc.M011698200. Epub 2001 Jun 21. J Biol Chem. 2001. PMID: 11423534
-
Synergistic activation of the type I adenylyl cyclase by Ca2+ and Gs-coupled receptors in vivo.J Biol Chem. 1994 Oct 14;269(41):25400-5. J Biol Chem. 1994. PMID: 7929237
-
Regulation of cyclic adenosine monophosphate synthesis in human ejaculated spermatozoa. II. The role of calcium and bicarbonate ions on the activation of adenylyl cyclase.Hum Reprod. 1992 Sep;7(8):1131-5. doi: 10.1093/oxfordjournals.humrep.a137807. Hum Reprod. 1992. PMID: 1328277
-
Soluble adenylyl cyclase of sea urchin spermatozoa.Biochim Biophys Acta. 2014 Dec;1842(12 Pt B):2621-8. doi: 10.1016/j.bbadis.2014.07.011. Epub 2014 Jul 23. Biochim Biophys Acta. 2014. PMID: 25064590 Free PMC article. Review.
-
Central role of soluble adenylyl cyclase and cAMP in sperm physiology.Biochim Biophys Acta. 2014 Dec;1842(12 Pt B):2610-20. doi: 10.1016/j.bbadis.2014.07.013. Epub 2014 Jul 24. Biochim Biophys Acta. 2014. PMID: 25066614 Free PMC article. Review.
Cited by
-
Calcium-sensing soluble adenylyl cyclase mediates TNF signal transduction in human neutrophils.J Exp Med. 2005 Aug 1;202(3):353-61. doi: 10.1084/jem.20050778. Epub 2005 Jul 25. J Exp Med. 2005. PMID: 16043520 Free PMC article.
-
CatSper1 required for evoked Ca2+ entry and control of flagellar function in sperm.Proc Natl Acad Sci U S A. 2003 Dec 9;100(25):14864-8. doi: 10.1073/pnas.2536658100. Epub 2003 Dec 1. Proc Natl Acad Sci U S A. 2003. PMID: 14657352 Free PMC article.
-
cAMP-dependent cell differentiation triggered by activated CRHR1 in hippocampal neuronal cells.Sci Rep. 2017 May 16;7(1):1944. doi: 10.1038/s41598-017-02021-7. Sci Rep. 2017. PMID: 28512295 Free PMC article.
-
Cyclic AMP produced inside mitochondria regulates oxidative phosphorylation.Cell Metab. 2009 Mar;9(3):265-76. doi: 10.1016/j.cmet.2009.01.012. Cell Metab. 2009. PMID: 19254571 Free PMC article.
-
Cyclic AMP dynamics in the pancreatic β-cell.Ups J Med Sci. 2012 Nov;117(4):355-69. doi: 10.3109/03009734.2012.724732. Epub 2012 Sep 13. Ups J Med Sci. 2012. PMID: 22970724 Free PMC article. Review.
References
-
- DasGupta, S., Mills, C. L. & Fraser, L. R. (1993) J. Reprod. Fertil. 99, 135–143. - PubMed
-
- Handrow, R. R., First, N. L. & Parrish, J. J. (1989) J. Exp. Zool. 252, 174–182. - PubMed
-
- Visconti, P. E., Bailey, J. L., Moore, G. D., Pan, D., Olds-Clarke, P. & Kopf, G. S. (1995) Development (Cambridge, U.K.) 121, 1129–1137. - PubMed
-
- Visconti, P. E., Stewart-Savage, J., Blasco, A., Battaglia, L., Miranda, P., Kopf, G. S. & Tezon, J. G. (1999) Biol. Reprod. 61, 76–84. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

