Distinct signaling pathways mediate stimulation of cell cycle progression and prevention of apoptotic cell death by estrogen in rat pituitary tumor PR1 cells
- PMID: 12960425
- PMCID: PMC284806
- DOI: 10.1091/mbc.e03-05-0303
Distinct signaling pathways mediate stimulation of cell cycle progression and prevention of apoptotic cell death by estrogen in rat pituitary tumor PR1 cells
Abstract
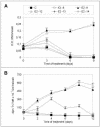
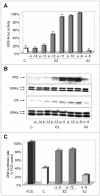
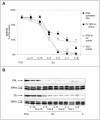
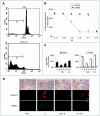
Estrogens control cell growth and viability in target cells via an interplay of genomic and extragenomic pathways not yet elucidated. Here, we show evidence that cell proliferation and survival are differentially regulated by estrogen in rat pituitary tumor PR1 cells. Pico- to femtomolar concentrations of 17beta-estradiol (E2) are sufficient to foster PR1 cell proliferation, whereas nanomolar concentrations of the same are needed to prevent cell death that occurs at a high rate in these cells in the absence of hormone. Activation of endogenous (PRL) or transfected estrogen-responsive genes occurs at the same, higher concentrations of E2 required to promote cell survival, whereas stimulation of cyclin D3 expression and DNA synthesis occur at lower E2 concentrations. Similarly, the pure antiestrogen ICI 182,780 inhibits estrogen response element-dependent trans-activation and cell death more effectively than cyclin-cdk activity, G1-S transition, or DNA synthesis rate. In antiestrogen-treated and/or estrogen-deprived cells, death is due predominantly to apoptosis. Estrogen-induced cell survival, but not E2-dependent cell cycle progression, can be prevented by an inhibitor of c-Src kinase or by blockade of the mitogen-activated protein kinase kinase/extracellular signal-regulated kinase signaling pathway. These data indicate the coexistence of two distinguishable estrogen signaling pathways in PR1 cells, characterized by different functions and sensitivity to hormones and antihormones.
Figures
Similar articles
-
High concentrations of bisphenol A induce cell growth and prolactin secretion in an estrogen-responsive pituitary tumor cell line.Toxicol Appl Pharmacol. 2000 Feb 1;162(3):161-5. doi: 10.1006/taap.1999.8840. Toxicol Appl Pharmacol. 2000. PMID: 10652244
-
Differential regulation by estrogens of growth and prolactin synthesis in pituitary cells suggests that only a small pool of estrogen receptors is required for growth.Proc Natl Acad Sci U S A. 1998 Mar 3;95(5):2325-30. doi: 10.1073/pnas.95.5.2325. Proc Natl Acad Sci U S A. 1998. PMID: 9482884 Free PMC article.
-
The antiestrogen ICI 182,780 inhibits proliferation of human breast cancer cells by interfering with multiple, sequential estrogen-regulated processes required for cell cycle completion.Mol Cell Endocrinol. 2000 Jul 25;165(1-2):199-209. doi: 10.1016/s0303-7207(00)00243-4. Mol Cell Endocrinol. 2000. PMID: 10940498
-
Nongenomic effects of estradiol vs. the birth control estrogen ethinyl estradiol on signaling and cell proliferation in pituitary tumor cells, and differences in the ability of R-equol to neutralize or enhance these effects.Steroids. 2021 Apr;168:108411. doi: 10.1016/j.steroids.2019.01.008. Epub 2019 May 25. Steroids. 2021. PMID: 31132367 Review.
-
Estrogens and the hair follicle.J Dtsch Dermatol Ges. 2004 Jun;2(6):412-23. doi: 10.1046/j.1439-0353.2004.04037.x. J Dtsch Dermatol Ges. 2004. PMID: 16281598 Review.
Cited by
-
Global analysis of estrogen receptor beta binding to breast cancer cell genome reveals an extensive interplay with estrogen receptor alpha for target gene regulation.BMC Genomics. 2011 Jan 14;12:36. doi: 10.1186/1471-2164-12-36. BMC Genomics. 2011. PMID: 21235772 Free PMC article.
-
Roles of dopamine 2 receptor isoforms and g proteins in ethanol regulated prolactin synthesis and lactotropic cell proliferation.PLoS One. 2012;7(9):e45593. doi: 10.1371/journal.pone.0045593. Epub 2012 Sep 18. PLoS One. 2012. PMID: 23029123 Free PMC article.
-
Effects of resveratrol on cell growth and prolactin synthesis in GH3 cells.Exp Ther Med. 2014 Apr;7(4):923-928. doi: 10.3892/etm.2014.1544. Epub 2014 Feb 13. Exp Ther Med. 2014. PMID: 24669252 Free PMC article.
-
Mediation of basic fibroblast growth factor-induced lactotropic cell proliferation by Src-Ras-mitogen-activated protein kinase p44/42 signaling.Endocrinology. 2005 Apr;146(4):1948-55. doi: 10.1210/en.2004-1448. Epub 2005 Jan 6. Endocrinology. 2005. PMID: 15637287 Free PMC article.
-
Selective estrogen receptor down-regulator and selective estrogen receptor modulators differentially regulate lactotroph proliferation.PLoS One. 2010 Apr 19;5(4):e10060. doi: 10.1371/journal.pone.0010060. PLoS One. 2010. PMID: 20419096 Free PMC article.
References
-
- Altucci, L., Addeo, R., Cicatiello, L., Dauvois, S., Parker, M.G., Truss, M., Beato, M., Sica, V., Bresciani, F., and Weisz, A. (1996). 17beta-Estradiol induces cyclin D1 gene transcription, p36D1-p34cdk4 complex activation and p105Rb phosphorylation during mitogenic stimulation of G(1)-arrested human breast cancer cells. Oncogene 12, 2315-2324. - PubMed
-
- Altucci, L., and Gronemeyer, H. (2001). Nuclear receptors in cell life and death. Trends Endocrinol. Metab. 12, 460-468. - PubMed
-
- Bacus, S.S., Gudkov, A.V., Lowe, M., Lyass, L., Yung, Y., Komarov, A.P., Keyomarsi, K., Yarden, Y., and Seger, R. (2001). Taxol-induced apoptosis depends on MAP kinase pathways (ERK and p38) and is independent of p53. Oncogene 20, 147-155. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

