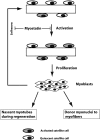
Myostatin negatively regulates satellite cell activation and self-renewal
- PMID: 12963705
- PMCID: PMC2172861
- DOI: 10.1083/jcb.200207056
Myostatin negatively regulates satellite cell activation and self-renewal
Abstract
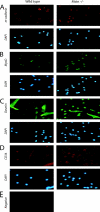
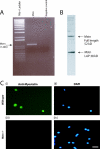
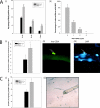
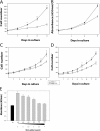
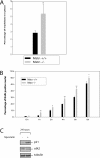
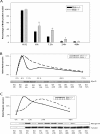
Satellite cells are quiescent muscle stem cells that promote postnatal muscle growth and repair. Here we show that myostatin, a TGF-beta member, signals satellite cell quiescence and also negatively regulates satellite cell self-renewal. BrdU labeling in vivo revealed that, among the Myostatin-deficient satellite cells, higher numbers of satellite cells are activated as compared with wild type. In contrast, addition of Myostatin to myofiber explant cultures inhibits satellite cell activation. Cell cycle analysis confirms that Myostatin up-regulated p21, a Cdk inhibitor, and decreased the levels and activity of Cdk2 protein in satellite cells. Hence, Myostatin negatively regulates the G1 to S progression and thus maintains the quiescent status of satellite cells. Immunohistochemical analysis with CD34 antibodies indicates that there is an increased number of satellite cells per unit length of freshly isolated Mstn-/- muscle fibers. Determination of proliferation rate suggests that this elevation in satellite cell number could be due to increased self-renewal and delayed expression of the differentiation gene (myogenin) in Mstn-/- adult myoblasts. Taken together, these results suggest that Myostatin is a potent negative regulator of satellite cell activation and thus signals the quiescence of satellite cells.
Figures

References
-
- Allen, R.E., C.J. Temm-Grove, S.M. Sheehan, and G. Rice. 1997. Skeletal muscle satellite cell cultures. Methods Cell Biol. 52:155–176. - PubMed
-
- Bischoff, R. 1989. Analysis of muscle regeneration using single myofibers in culture. Med. Sci. Sports Exerc. 21:S164–S172. - PubMed
-
- Bogdanovich, S., T.O. Krag, E.R. Barton, L.D. Morris, L.A. Whittemore, R.S. Ahima, and T.S. Khurana. 2002. Functional improvement of dystrophic muscle by myostatin blockade. Nature. 420:418–421. - PubMed
-
- Carlson, C.J., F.W. Booth, and S.E. Gordon. 1999. Skeletal muscle myostatin mRNA expression is fiber-type specific and increases during hindlimb unloading. Am. J. Physiol. 277:R601–R606. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

