Phase II study of cisplatin, ifosfamide, and irinotecan with rhG-CSF support in patients with stage IIIb and IV non-small-cell lung cancer
- PMID: 12966417
- PMCID: PMC2376953
- DOI: 10.1038/sj.bjc.6601230
Phase II study of cisplatin, ifosfamide, and irinotecan with rhG-CSF support in patients with stage IIIb and IV non-small-cell lung cancer
Abstract
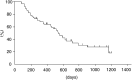
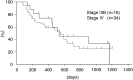
A phase II study of cisplatin, ifosfamide, and irinotecan with recombinant human granulocyte colony stimulating factor (rhG-CSF) support was conducted in previously untreated patients with stage IIIB or IV non-small-cell lung cancer (NSCLC). Between June 1998 and August 2001, 50 patients were registered in this phase II study. Cisplatin (20 mg m(-2)) and ifosfamide (1.5 g m(-2)) were administered on days 1-4 and irinotecan (60 mg m(-2)) was given on days 1, 8, and 15, respectively. This regimen was repeated every 4 weeks. rhG-CSF was administered subcutaneously at a dose of 50 microg m(-2) on days 5-18 except on the days of irinotecan treatment. In total, 49 patients were assessable for toxicity and response and 50 for survival. In all, 33, patients (67.3%; 95% confidence interval 57.4-77.2%) achieved an objective response. The median response duration was 192 days and the median time to progression for 49 patients was 170 days. The median survival time was 540 days with 1- and 2-year survival rates of 63.5 and 30.7%, respectively. Grade 3 or 4 neutropenia and thrombocytopenia developed in 63.3 and 38.8% of the patients, respectively. In conclusion, the combination of cisplatin, ifosfamide, and irinotecan with rhG-CSF support was highly effective for the treatment of stage IIIB or IV NSCLC with acceptable toxicities.
Figures
Similar articles
-
Phase I/II study of cisplatin, ifosfamide and irinotecan with rhG-CSF support in patients with stage IIIB and IV non-small-cell lung cancer.Cancer Chemother Pharmacol. 2000;45(4):279-83. doi: 10.1007/s002800050041. Cancer Chemother Pharmacol. 2000. PMID: 10755315 Clinical Trial.
-
Phase I study of cisplatin, ifosfamide and irinotecan with rhG-CSF support in advanced non-small cell lung cancer.Oncology. 1999;56(4):301-7. doi: 10.1159/000011982. Oncology. 1999. PMID: 10343194 Clinical Trial.
-
Phase I study of carboplatin, irinotecan and docetaxel on a divided schedule with recombinant human granulocyte colony stimulating factor support in patients with stage IIIB or IV non-small cell lung cancer.Anticancer Drugs. 2002 Jun;13(5):505-9. doi: 10.1097/00001813-200206000-00009. Anticancer Drugs. 2002. PMID: 12045462 Clinical Trial.
-
Combination of cisplatin, ifosfamide, and irinotecan with rhG-CSF support for the treatment of refractory or relapsed small-cell lung cancer.Oncology. 2000 Aug;59(2):105-9. doi: 10.1159/000012145. Oncology. 2000. PMID: 10971167 Clinical Trial.
-
Establishment of the standard regimen for non-small-cell lung cancer in Japan.Oncology (Williston Park). 2001 Jan;15(1 Suppl 1):13-8. Oncology (Williston Park). 2001. PMID: 11221016 Review.
Cited by
-
rhG-CSF is associated with an increased risk of metastasis in NSCLC patients following postoperative chemotherapy.BMC Cancer. 2022 Jul 7;22(1):741. doi: 10.1186/s12885-022-09850-4. BMC Cancer. 2022. PMID: 35799161 Free PMC article.
-
The Effects of GCSF Primary Prophylaxis on Survival Outcomes and Toxicity in Patients with Advanced Non-Small Cell Lung Cancer on First-Line Chemoimmunotherapy: A Sub-Analysis of the Spinnaker Study.Int J Mol Sci. 2023 Jan 16;24(2):1746. doi: 10.3390/ijms24021746. Int J Mol Sci. 2023. PMID: 36675262 Free PMC article.
-
Chemotherapy of lung cancer: A global perspective of the role of ifosfamide.Transl Lung Cancer Res. 2012 Mar;1(1):61-71. doi: 10.3978/j.issn.2218-6751.2011.12.02. Transl Lung Cancer Res. 2012. PMID: 25806156 Free PMC article.
References
-
- Costanza JJ, Gagliano R, Loukas D, Panttiere FJ, Hokanson JA (1978) Ifosfamide in recurrent or disseminated lung cancer. Cancer 41: 1751–1719 - PubMed
-
- Fujita A, Ohkubo T, Hoshino H, Takabatake H, Tagaki S, Sekine K (2002) Phase I study of carboplatin, irinotecan and docetaxel on a divided schedule with recombinant human granulocyte colony stimulating factor (rhG-CSF) support in patients with stage IIIB or IV non-small cell lung cancer. Anti-Cancer Drugs 13: 505–509 - PubMed
-
- Fujita A, Takabatake H, Tagaki S, Sekine K (1999) Phase I study of cisplatin, ifosfamide and irinotecan with rhG-CSF support in advanced non-small cell lung cancer. Oncology 56: 307–307 - PubMed
-
- Fujita A, Takabatake H, Tagaki S, Sekine K (2000a) Combination chemotherapy of cisplatin, ifosfamide and irinotecan with rhG-CSF support in patients with brain metastases from non-small cell lung cancer. Oncology 59: 291–295 - PubMed
-
- Fujita A, Takabatake H, Tagaki S, Sekine K (2000b) Phase I study of carboplatin, docetaxel and irinotecan with recombinant human granulocyte colony stimulating factor support in patients with advanced non-small cell lung cancer. Anti-Cancer Drugs 11: 821–824 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical