Nonneurotropic adenovirus: a vector for gene transfer to the brain and gene therapy of neurological disorders
- PMID: 12968530
- PMCID: PMC2902245
- DOI: 10.1016/s0074-7742(03)01001-8
Nonneurotropic adenovirus: a vector for gene transfer to the brain and gene therapy of neurological disorders
Figures
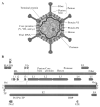
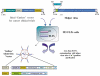
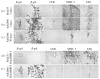

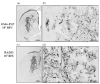
Similar articles
-
Immune responses to adenovirus and adeno-associated vectors used for gene therapy of brain diseases: the role of immunological synapses in understanding the cell biology of neuroimmune interactions.Curr Gene Ther. 2007 Oct;7(5):347-60. doi: 10.2174/156652307782151498. Curr Gene Ther. 2007. PMID: 17979681 Free PMC article. Review.
-
Biology of adenovirus and its use as a vector for gene therapy.Hum Gene Ther. 2004 Nov;15(11):1022-33. doi: 10.1089/hum.2004.15.1022. Hum Gene Ther. 2004. PMID: 15610603 Review. No abstract available.
-
Adenovirus-mediated gene transfer to the brain: methodological assessment.J Neurosci Methods. 1997 Jan;71(1):77-84. doi: 10.1016/s0165-0270(96)00128-8. J Neurosci Methods. 1997. PMID: 9125377 Review.
-
Adenovirus vectors and subviral particles for protein and peptide delivery.Curr Gene Ther. 2012 Oct;12(5):362-73. doi: 10.2174/156652312802762563. Curr Gene Ther. 2012. PMID: 22845888 Review.
-
Adenovirus-mediated gene transfer to liver.Adv Drug Deliv Rev. 2001 Mar 1;46(1-3):205-9. doi: 10.1016/s0169-409x(00)00125-3. Adv Drug Deliv Rev. 2001. PMID: 11259841 Review. No abstract available.
Cited by
-
Viral strategies for studying the brain, including a replication-restricted self-amplifying delta-G vesicular stomatis virus that rapidly expresses transgenes in brain and can generate a multicolor golgi-like expression.J Comp Neurol. 2009 Oct 20;516(6):456-81. doi: 10.1002/cne.22131. J Comp Neurol. 2009. PMID: 19672982 Free PMC article.
-
Regulatable gene expression systems for gene therapy applications: progress and future challenges.Mol Ther. 2005 Aug;12(2):189-211. doi: 10.1016/j.ymthe.2005.03.022. Mol Ther. 2005. PMID: 15946903 Free PMC article. Review.
-
Regulatable gene expression systems for gene therapy.Curr Gene Ther. 2006 Aug;6(4):421-38. doi: 10.2174/156652306777934829. Curr Gene Ther. 2006. PMID: 16918333 Review.
-
Engineered mitochondrial ferritin as a magnetic resonance imaging reporter in mouse olfactory epithelium.PLoS One. 2013 Aug 30;8(8):e72720. doi: 10.1371/journal.pone.0072720. eCollection 2013. PLoS One. 2013. PMID: 24023635 Free PMC article.
-
Human gene therapy and imaging in neurological diseases.Eur J Nucl Med Mol Imaging. 2005 Dec;32 Suppl 2(Suppl 2):S358-83. doi: 10.1007/s00259-005-1960-3. Eur J Nucl Med Mol Imaging. 2005. PMID: 16328505 Free PMC article. Review.
References
-
- Acsadi G, Jani A, et al. Cultured human myoblasts and myotubes show markedly different transducibility by replication-defective adenovirus recombinants. Gene Ther. 1994a;1(5):338–340. - PubMed
-
- Acsadi G, Jani A, et al. A differential efficiency of adenovirus-mediated in vivo gene transfer into skeletal muscle cells of different maturity. Hum. Mol. Genet. 1994b;3(4):579–584. - PubMed
-
- Ailles LE, Naldini L. HIV-1-derived lentiviral vectors. Curr. Top. Microbiol. Immunol. 2002;261:31–52. - PubMed
-
- Akli S, Caillaud C, et al. Transfer of a foreign gene into the brain using adenovirus vectors. Nat. Genet. 1993;3(3):224–228. - PubMed
aReferences to Table II
-
- Abe T, Wakimoto H, et al. Intra-arterial delivery of p53-containing adenoviral vector into experimental brain tumors. Cancer Gene Ther. 2002;9(3):228–235. - PubMed
-
- Adachi Y, Tamiya T, et al. Experimental gene therapy for brain tumors using adenovirus-mediated transfer of cytosine deaminase gene and uracil phosphoribosyltransferase gene with 5-fluorocytosine. Hum. Gene Ther. 2000;11(1):77–89. - PubMed
-
- Adachi Y, Chandrasekar N, et al. Suppression of glioma invasion and growth by adenovirus-mediated delivery of a bicistronic construct containing antisense uPAR and sense p16 gene sequences. Oncogene. 2002;21(1):87–95. - PubMed
-
- Akli S, Caillaud C, et al. Transfer of a foreign gene into the brain using adenovirus vectors. Nat. Genet. 1993;3(3):224–228. - PubMed
-
- Aoki M, Tamatani M, et al. Hypothermic treatment restores glucose regulated protein 78 (GRP78) expression in ischemic brain. Brain Res. Mol. Brain Res. 2001;95(1–2):117–128. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
