Access of antibody molecules to the conserved coreceptor binding site on glycoprotein gp120 is sterically restricted on primary human immunodeficiency virus type 1
- PMID: 12970440
- PMCID: PMC228502
- DOI: 10.1128/jvi.77.19.10557-10565.2003
Access of antibody molecules to the conserved coreceptor binding site on glycoprotein gp120 is sterically restricted on primary human immunodeficiency virus type 1
Abstract
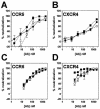
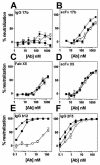
Anti-human immunodeficiency virus type 1 (HIV-1) antibodies whose binding to gp120 is enhanced by CD4 binding (CD4i antibodies) are generally considered nonneutralizing for primary HIV-1 isolates. However, a novel CD4i-specific Fab fragment, X5, has recently been found to neutralize a wide range of primary isolates. To investigate the precise nature of the extraordinary neutralizing ability of Fab X5, we evaluated the abilities of different forms (immunoglobulin G [IgG], Fab, and single-chain Fv) of X5 and other CD4i monoclonal antibodies to neutralize a range of primary HIV-1 isolates. Our results show that, for a number of isolates, the size of the neutralizing agent is inversely correlated with its ability to neutralize. Thus, the poor ability of CD4i-specific antibodies to neutralize primary isolates is due, at least in part, to steric factors that limit antibody access to the gp120 epitopes. Studies of temperature-regulated neutralization or fusion-arrested intermediates suggest that the steric effects are important in limiting the binding of IgG to the viral envelope glycoproteins after HIV-1 has engaged CD4 on the target cell membrane. The results identify hurdles in using CD4i epitopes as targets for antibody-mediated neutralization in vaccine design but also indicate that the CD4i regions could be efficiently targeted by small molecule entry inhibitors.
Figures
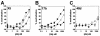

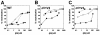
References
-
- Barbas, C. F., III, D. R. Burton, J. K. Scott, and G. J. Silverman. 2001. Phage display: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
-
- Binley, J. M., R. W. Sanders, B. Clas, N. Schuelke, A. Master, Y. Guo, F. Kajumo, D. J. Anselma, P. J. Maddon, W. C. Olson, and J. P. Moore. 2000. A recombinant human immunodeficiency virus type 1 envelope glycoprotein complex stabilized by an intermolecular disulfide bond between the gp120 and gp41 subunits is an antigenic mimic of the trimeric virion-associated structure. J. Virol. 74:627-643. - PMC - PubMed
-
- Burton, D. R., J. Pyati, R. Koduri, S. J. Sharp, G. B. Thornton, P. W. Parren, L. S. Sawyer, R. M. Hendry, N. Dunlop, P. L. Nara, et al. 1994. Efficient neutralization of primary isolates of HIV-1 by a recombinant human monoclonal antibody. Science 266:1024-1027. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- AI42848/AI/NIAID NIH HHS/United States
- R37 AI024755/AI/NIAID NIH HHS/United States
- R01 AI041851/AI/NIAID NIH HHS/United States
- AI31783/AI/NIAID NIH HHS/United States
- R01 AI031783/AI/NIAID NIH HHS/United States
- AI39420/AI/NIAID NIH HHS/United States
- R01 AI039420/AI/NIAID NIH HHS/United States
- AI41851/AI/NIAID NIH HHS/United States
- P30 AI042848/AI/NIAID NIH HHS/United States
- R37 AI033292/AI/NIAID NIH HHS/United States
- R01 AI033292/AI/NIAID NIH HHS/United States
- R01 AI045357/AI/NIAID NIH HHS/United States
- AI33292/AI/NIAID NIH HHS/United States
- AI49566/AI/NIAID NIH HHS/United States
- AI45357/AI/NIAID NIH HHS/United States
- AI24755/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

