Heat sensitivity in a bentgrass variant. Failure to accumulate a chloroplast heat shock protein isoform implicated in heat tolerance
- PMID: 12970497
- PMCID: PMC196608
- DOI: 10.1104/pp.102.018309
Heat sensitivity in a bentgrass variant. Failure to accumulate a chloroplast heat shock protein isoform implicated in heat tolerance
Abstract
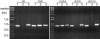
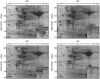
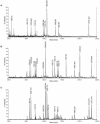
Two variants of creeping bentgrass (Agrostis stolonifera cv palustris), developed using tissue culture, have been used to determine the roles of chloroplast-localized small heat shock proteins (CP-sHSPs) in heat tolerance. Results from previous research indicate that the heat-tolerant variant expressed two additional CP-sHSP isoforms not expressed in the heat-sensitive variant, that accumulation of the additional CP-sHSP isoforms was genetically linked to thermotolerance, and that the presence of the additional isoforms in the heat-tolerant variant provided greater protection to photosystem II during heat stress. To determine the basis of the differential expression, we isolated the genes encoding the CP-sHSPs from both variants and characterized their structure and expression. Two genes, ApHsp26.2 and ApHsp26.7a, were isolated from the heat-tolerant variant, and three genes, ApHsp26.2m, ApHsp26.8, and ApHsp26.7b, were isolated from the heat-sensitive variant. The sequence of ApHsp26.2m from the heat-sensitive variant was identical to ApHsp26.2, except for a point mutation that generated a premature stop codon. Therefore, the protein product of ApHsp26.2m did not accumulate in the heat-sensitive line. Mass spectrometry analysis confirmed that ApHsp26.2 encoded for the CP-sHSP isoforms unique to the heat-tolerant variant. An identical mutation was detected in one of the three parental lines used to develop the creeping bentgrass variants. This suggests that ApHsp26.2m was inherited from this parent and did not arise from a mutation that occurred during tissue culture. The presence of two isoforms encoded by the same gene might be due to differential processing of the N-terminal amino acids during or after import into the chloroplast.
Figures

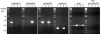

References
-
- Downs CA, Heckathorn SA, Bryan JK, Coleman JS (1998) The methionine-rich low-molecular-weight chloroplast heat-shock protein: evolutionary conservation and accumulation in relation to thermotolerance. Am J Bot 85: 175-183 - PubMed
-
- Downs CA, Ryan SL, Heckathorn SA (1999) The chloroplast small heat-shock protein: evidence for a general role in protecting photosystem II against oxidative stress and photoinhibition. J Plant Physiol 155: 488-496
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Miscellaneous

