Normal B cell homeostasis requires B cell activation factor production by radiation-resistant cells
- PMID: 12975458
- PMCID: PMC2194202
- DOI: 10.1084/jem.20030789
Normal B cell homeostasis requires B cell activation factor production by radiation-resistant cells
Abstract
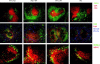
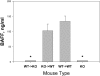
The cellular source of B cell activation factor (BAFF) required for peripheral B cell survival/maturation is unknown. To determine the nature of BAFF-producing cells we established and analyzed reciprocal bone marrow (BM) chimeras with wild-type (WT) and BAFF-deficient mice. The results revealed that BAFF production by radiation-resistant stromal cells is completely sufficient to provide a necessary signal for B cell survival/maturation, as BAFF-/- BM cells transferred into lethally irradiated WT mice gave rise to normal numbers of follicular (FO) and marginal zone (MZ) B cell subpopulations. On the other hand, transfer of WT BM into BAFF-/- lethally irradiated mice resulted only in minimal reconstitution of mature FO B cells and no restoration of MZ B cells. Thus, in the absence of BAFF+/+ stromal cells, BAFF production by BM-derived cells, presumably by macrophages, dendritic cells, and/or neutrophils, was not at all sufficient to support normal B cell homeostasis. Interestingly, immunization of both types of chimeras stimulated high levels of antigen-specific antibody secretion, indicating that either stromal cell- or hematopoietic cell-derived BAFF is sufficient for B cell antibody responses.
Figures




References
-
- Shu, H., W. Hu, and H. Johnson. 1999. TALL-1 is a novel member of the TNF family that is down-regulated by mitogens. J. Leukoc. Biol. 65:680–683. - PubMed
-
- Mukhopadhyay, A., J. Ni, Y. Zhai, G.-L. Yu, and B.B. Aggarwal. 1999. Identification and characterization of a novel cytokine, THANK, a TNF homologue that activates apoptosis, nuclear factor-kappa B, and c-Jun NH2-terminal kinase. J. Biol. Chem. 274:15978–15981. - PubMed
-
- Moore, P.A., O. Belvedere, A. Orr, K. Pieri, D.W. LaFleur, P. Feng, D. Soppet, M. Charters, R. Gentz, D. Parmelee, et al. 1999. BLyS: member of the tumor necrosis factor family and B lymphocyte stimulator. Science. 285:260–263. - PubMed
-
- Thompson, J.S., P. Schneider, S.L. Kalled, L. Wang, E.A. Lefevre, T.G. Cachero, F. MacKay, S.A. Bixler, M. Zafari, Z.Y. Liu, et al. 2000. BAFF binds to the tumor necrosis factor receptor–like molecule B cell maturation antigen and is important for maintaining the peripheral B cell population. J. Exp. Med. 192:129–135. - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

