The 9E3 protein: immunolocalization in vivo and evidence for multiple forms in culture
- PMID: 1325984
- PMCID: PMC2937011
- DOI: 10.1242/jcs.101.3.701
The 9E3 protein: immunolocalization in vivo and evidence for multiple forms in culture
Abstract
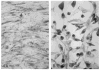
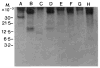
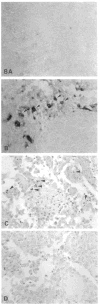
The avian gene 9E3/CEF4 belongs to a group of genes whose products are highly conserved and are homologous to inflammatory mediators. These genes, sometimes referred to as the gro family, are also expressed upon wounding or serum-stimulation of quiescent cells, suggesting that they may be important in aspects of growth and/or wound healing. We have used an antibody to the product of the 9E3 gene to show for the first time the distribution in vivo of the protein of one of these genes. The polyclonal antibody was produced against a synthetic peptide, [Cys76], 9E3, (77-103), located at the carboxy end of the molecule. The specificity of the antibody was determined by transfection of the 9E3 cDNA into Cos 7 cells, which do not express this gene. Moreover, despite the high homology between 9E3 and IL-8, the antibody did not crossreact with this molecule. The antibody was used to immuno-precipitate the protein from cultured normal and RSV-transformed chick embryo fibroblasts (CEFs) and to determine its distribution in tissues of newly hatched chicks. The staining was abundant in the cells and extracellular matrix (ECM) of connective tissue and other tissues of mesenchymal origin, such as bone and tendon. Most cells in the granulation tissue of wounds stained, some more intensely than others; the ECM also stained, especially in areas of scar tissue where collagen is abundant. In RSV-induced tumors, the protein was absent except in necrotic areas where a few cells--potentially macrophages--stained. In general, as expected, the protein was present in the cells and tissues that expressed the mRNA, but there were exceptions.(ABSTRACT TRUNCATED AT 250 WORDS)
Figures





References
-
- Appella E, Matushima K, Oppenhelm JJ, Yoshlmura T, Leonard EJ, Clore GM, Gronenborn AM. Determination of the primary and secondary structure of NAP-1/IL-8 and a monocyte chemoattractant protein, MCP-l/MCAF. Prog Clin Biol Res. 1990;349:405–417. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

