Decoding of synaptic voltage waveforms by specific classes of recombinant high-threshold Ca(2+) channels
- PMID: 14500770
- PMCID: PMC2343577
- DOI: 10.1113/jphysiol.2003.051110
Decoding of synaptic voltage waveforms by specific classes of recombinant high-threshold Ca(2+) channels
Abstract
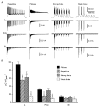
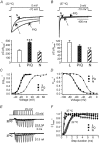
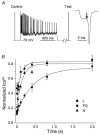
Studies suggest that the preferential role of L-type voltage-sensitive Ca(2+) channels (VSCCs) in coupling strong synaptic stimulation to transcription is due to their selective activation of local chemical events. However, it is possible that selective activation of the L-type channel by specific voltage waveforms also makes a contribution. To address this issue we have examined the response of specific Ca(2+) channel types to simulated complex voltage waveforms resembling those encountered during synaptic plasticity (gamma and theta firing frequency). L-, P/Q- and N-type VSCCs (alpha1C, alpha1A, alpha1B/beta1B/alpha2delta, respectively) were all similarly activated by brief action potential (AP) waveforms or sustained step depolarization. When complex waveforms containing large excitatory postsynaptic potentials (EPSPs), APs and spike accommodation were applied under voltage clamp we found that the integrated L-type VSCC current was approximately three times larger than that produced by the P/Q- or N-type Ca(2+) channels (gamma frequency 1 s stimulation). For P/Q- or N-type channels the complex waveforms led to a smaller current than that expected from the response to a simple 1 s step depolarization to 0 or +20 mV. EPSPs present in the waveforms favoured the inactivation of P/Q- and N-type channels. In contrast, activation of the L-type channel was dependent on both EPSP- and AP-mediated depolarization. Expression of P/Q-type channels with reduced voltage-dependent inactivation (alpha1A/beta2A/alpha2delta) or the use of hyperpolarized intervals between AP stimuli greatly increased their response to complex voltage stimuli. We propose that in response to complex synaptic voltage waveforms P/Q- and N-type channels can undergo selective voltage-dependent inactivation leading to a Ca(2+) current mediated predominantly by L-type channels.
Figures




Similar articles
-
Voltage-dependent calcium channels.Gen Physiol Biophys. 2005 Jun;24 Suppl 1:1-78. Gen Physiol Biophys. 2005. PMID: 16096350 Review.
-
Voltage-dependent calcium channels in the rat retina: involvement in NMDA-stimulated influx of calcium.Exp Eye Res. 2001 Apr;72(4):393-401. doi: 10.1006/exer.2000.0968. Exp Eye Res. 2001. PMID: 11273667
-
A novel molecular inactivation determinant of voltage-gated CaV1.2 L-type Ca2+ channel.Neuroscience. 2006;139(4):1275-87. doi: 10.1016/j.neuroscience.2006.01.028. Epub 2006 Mar 14. Neuroscience. 2006. PMID: 16533566
-
High-voltage-activated calcium channels in Muller cells acutely isolated from tiger salamander retina.Glia. 2005 Jan 15;49(2):259-74. doi: 10.1002/glia.20113. Glia. 2005. PMID: 15472989
-
[P/Q-type voltage-dependent calcium channels in neurological disease].Neurologia. 2007 Oct;22(8):511-6. Neurologia. 2007. PMID: 17573560 Review. Spanish.
Cited by
-
A Rich Conformational Palette Underlies Human CaV2.1-Channel Availability.bioRxiv [Preprint]. 2024 Sep 30:2024.09.27.615501. doi: 10.1101/2024.09.27.615501. bioRxiv. 2024. Update in: Nat Commun. 2025 Apr 23;16(1):3815. doi: 10.1038/s41467-025-58884-2. PMID: 39464068 Free PMC article. Updated. Preprint.
-
Exploring the dominant role of Cav1 channels in signalling to the nucleus.Biosci Rep. 2012 Dec 20;33(1):97-101. doi: 10.1042/BSR20120099. Biosci Rep. 2012. PMID: 23088728 Free PMC article. Review.
-
Ca(V)1 and Ca(V)2 channels engage distinct modes of Ca(2+) signaling to control CREB-dependent gene expression.Cell. 2012 May 25;149(5):1112-24. doi: 10.1016/j.cell.2012.03.041. Cell. 2012. PMID: 22632974 Free PMC article.
-
Facilitation of L-type Ca2+ channels in dendritic spines by activation of beta2 adrenergic receptors.J Neurosci. 2004 Sep 29;24(39):8416-27. doi: 10.1523/JNEUROSCI.1677-04.2004. J Neurosci. 2004. PMID: 15456814 Free PMC article.
-
Calcium signaling in synapse-to-nucleus communication.Cold Spring Harb Perspect Biol. 2011 Nov 1;3(11):a004564. doi: 10.1101/cshperspect.a004564. Cold Spring Harb Perspect Biol. 2011. PMID: 21791697 Free PMC article. Review.
References
-
- Bading H, Ginty DD, Greenberg ME. Regulation of gene expression in hippocampal neurons by distinct calcium signaling pathways. Science. 1993;260:181–186. - PubMed
-
- Barnes-Davies M, Owens S, Forsythe ID. Calcium channels triggering transmitter release in the rat medial superior olive. Hear Res. 2001;162:134–145. - PubMed
-
- Bourinet E, Soong TW, Sutton K, Slaymaker S, Mathews E, Monteil A, Zamponi GW, Nargeot J, Snutch TP. Splicing of alpha 1A subunit gene generates phenotypic variants of P- and Q-type calcium channels. Nat Neurosci. 1999;2:407–415. - PubMed
-
- Christie BR, Eliot LS, Ito K, Miyakawa H, Johnston D. Different Ca2+ channels in soma and dendrites of hippocampal pyramidal neurons mediate spike-induced Ca2+ influx. J Neurophysiol. 1995a;73:2553–2557. - PubMed
-
- Christie BR, Stellwagen D, Abraham WC. Reduction of the threshold for long-term potentiation by prior theta- frequency synaptic activity. Hippocampus. 1995b;5:52–59. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

