A carboxyl-terminal interaction of lamin B1 is dependent on the CAAX endoprotease Rce1 and carboxymethylation
- PMID: 14504265
- PMCID: PMC2173957
- DOI: 10.1083/jcb.200303113
A carboxyl-terminal interaction of lamin B1 is dependent on the CAAX endoprotease Rce1 and carboxymethylation
Abstract
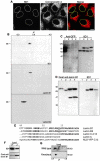
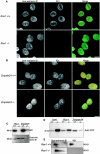
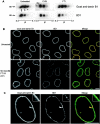
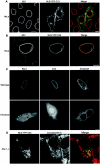
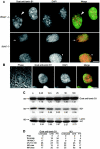
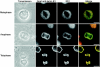
The mammalian nuclear lamina protein lamin B1 is posttranslationally modified by farnesylation, endoproteolysis, and carboxymethylation at a carboxyl-terminal CAAX motif. In this work, we demonstrate that the CAAX endoprotease Rce1 is required for lamin B1 endoproteolysis, demonstrate an independent pool of proteolyzed but nonmethylated lamin B1, as well as fully processed lamin B1, in interphase nuclei, and show a role for methylation in the organization of lamin B1 into domains of the nuclear lamina. Deficiency in the endoproteolysis or methylation of lamin B1 results in loss of integrity and deformity of the nuclear lamina. These data show that the organization of the nuclear envelope and lamina is dependent on a mechanism involving the methylation of lamin B1, and they identify a potential mechanism of laminopathy involving a B-type lamin.
Figures

References
-
- Adjei, A.A., J.N. Davis, C. Erlichman, P.A. Svingen, and S.H. Kaufmann. 2000. Comparison of potential markers of farnesyltransferase inhibition. Clin. Cancer Res. 6:2318–2325. - PubMed
-
- Bergo, M.O., G.K. Leung, P. Ambroziak, J.C. Otto, P.J. Casey, and S.G. Young. 2000. Targeted inactivation of the isoprenylcysteine carboxyl methyltransferase gene causes mislocalization of K-Ras in mammalian cells. J. Biol. Chem. 275:17605–17610. - PubMed
-
- Bergo, M.O., G.K. Leung, P. Ambroziak, J.C. Otto, P.J. Casey, A.Q. Gomes, M.C. Seabra, and S.G. Young. 2001. Isoprenylcysteine carboxyl methyltransferase deficiency in mice. J. Biol. Chem. 276:5841–5845. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

