Phosphatidylinositol-4,5-bisphosphate, PIP2, controls KCNQ1/KCNE1 voltage-gated potassium channels: a functional homology between voltage-gated and inward rectifier K+ channels
- PMID: 14532114
- PMCID: PMC213780
- DOI: 10.1093/emboj/cdg526
Phosphatidylinositol-4,5-bisphosphate, PIP2, controls KCNQ1/KCNE1 voltage-gated potassium channels: a functional homology between voltage-gated and inward rectifier K+ channels
Abstract
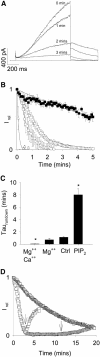
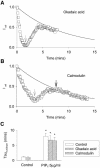
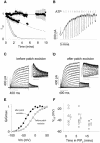
Phosphatidylinositol-4,5-bisphosphate (PIP(2)) is a major signaling molecule implicated in the regulation of various ion transporters and channels. Here we show that PIP(2) and intracellular MgATP control the activity of the KCNQ1/KCNE1 potassium channel complex. In excised patch-clamp recordings, the KCNQ1/KCNE1 current decreased spontaneously with time. This rundown was markedly slowed by cytosolic application of PIP(2) and fully prevented by application of PIP(2) plus MgATP. PIP(2)-dependent rundown was accompanied by acceleration in the current deactivation kinetics, whereas the MgATP-dependent rundown was not. Cytosolic application of PIP(2) slowed deactivation kinetics and also shifted the voltage dependency of the channel activation toward negative potentials. Complex changes in the current characteristics induced by membrane PIP(2) was fully restituted by a model originally elaborated for ATP-regulated two transmembrane-domain potassium channels. The model is consistent with stabilization by PIP(2) of KCNQ1/KCNE1 channels in the open state. Our data suggest a striking functional homology between a six transmembrane-domain voltage-gated channel and a two transmembrane-domain ATP-gated channel.
Figures




References
-
- Barhanin J., Lesage,F., Guillemare,E., Fink,M., Lazdunski,M. and Romey,G. (1996) K(V)LQT1 and lsK (minK) proteins associate to form the I(Ks) cardiac potassium current. Nature, 384, 78–80. - PubMed
-
- Baró I. and Escande,D. (1989) A long lasting Ca2+-activated outward current in guinea-pig atrial myocytes. Pflugers Arch., 415, 63–71. - PubMed
-
- Baukrowitz T and Fakler,B. (2000) K(ATP) channels: linker between phospholipid metabolism and excitability. Biochem. Pharmacol., 60, 735–740. - PubMed
-
- Baukrowitz T., Schulte,U., Oliver,D., Herlitze,S., Krauter,T., Tucker,S.J. Ruppersberg,J.P. and Fakler,B. (1998) PIP2 and PIP as determinants for ATP inhibition of KATP channels. Science, 282, 1141–1144. - PubMed
-
- Becq F. (1996) Ionic channel rundown in excised membrane patches. Biochim. Biophys Acta, 1286, 53–63. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

