AIP is a mitochondrial import mediator that binds to both import receptor Tom20 and preproteins
- PMID: 14557246
- PMCID: PMC2173431
- DOI: 10.1083/jcb.200305051
AIP is a mitochondrial import mediator that binds to both import receptor Tom20 and preproteins
Abstract
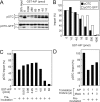
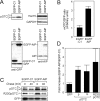
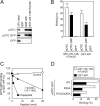
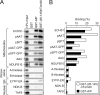
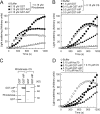
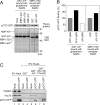
Most mitochondrial preproteins are maintained in a loosely folded import-competent conformation by cytosolic chaperones, and are imported into mitochondria by translocator complexes containing a preprotein receptor, termed translocase of the outer membrane of mitochondria (Tom) 20. Using two-hybrid screening, we identified arylhydrocarbon receptor-interacting protein (AIP), an FK506-binding protein homologue, interacting with Tom20. The extreme COOH-terminal acidic segment of Tom20 was required for interaction with tetratricopeptide repeats of AIP. An in vitro import assay indicated that AIP prevents preornithine transcarbamylase from the loss of import competency. In cultured cells, overexpression of AIP enhanced preornithine transcarbamylase import, and depletion of AIP by RNA interference impaired the import. An in vitro binding assay revealed that AIP specifically binds to mitochondrial preproteins. Formation of a ternary complex of Tom20, AIP, and preprotein was observed. Hsc70 was also found to bind to AIP. An aggregation suppression assay indicated that AIP has a chaperone-like activity to prevent substrate proteins from aggregation. These results suggest that AIP functions as a cytosolic factor that mediates preprotein import into mitochondria.
Figures



Similar articles
-
Functional analysis of human metaxin in mitochondrial protein import in cultured cells and its relationship with the Tom complex.Biochem Biophys Res Commun. 2000 Oct 5;276(3):1028-34. doi: 10.1006/bbrc.2000.3589. Biochem Biophys Res Commun. 2000. PMID: 11027586
-
Tom22 is a multifunctional organizer of the mitochondrial preprotein translocase.Nature. 1999 Sep 30;401(6752):485-9. doi: 10.1038/46802. Nature. 1999. PMID: 10519552
-
Tom5 functionally links mitochondrial preprotein receptors to the general import pore.Nature. 1997 Jul 10;388(6638):195-200. doi: 10.1038/40663. Nature. 1997. PMID: 9217162
-
Mitochondrial preprotein translocases as dynamic molecular machines.FEMS Yeast Res. 2006 Sep;6(6):849-61. doi: 10.1111/j.1567-1364.2006.00134.x. FEMS Yeast Res. 2006. PMID: 16911507 Review.
-
Protein import into mitochondria of Neurospora crassa.Fungal Genet Biol. 2002 Jul;36(2):85-90. doi: 10.1016/S1087-1845(02)00018-X. Fungal Genet Biol. 2002. PMID: 12081461 Review.
Cited by
-
Biological Role and Mechanism of Lipid Metabolism Reprogramming Related Gene ECHS1 in Cancer.Technol Cancer Res Treat. 2022 Jan-Dec;21:15330338221140655. doi: 10.1177/15330338221140655. Technol Cancer Res Treat. 2022. PMID: 36567598 Free PMC article. Review.
-
Cytosolic factor- and TOM-independent import of C-tail-anchored mitochondrial outer membrane proteins.EMBO J. 2006 Dec 13;25(24):5635-47. doi: 10.1038/sj.emboj.7601438. Epub 2006 Nov 16. EMBO J. 2006. PMID: 17110923 Free PMC article.
-
Survivin and IAP proteins in cell-death mechanisms.Biochem J. 2010 Sep 1;430(2):199-205. doi: 10.1042/BJ20100814. Biochem J. 2010. PMID: 20704571 Free PMC article. Review.
-
Common ground for protein translocation: access control for mitochondria and chloroplasts.Nat Rev Mol Cell Biol. 2011 Jan;12(1):48-59. doi: 10.1038/nrm3027. Epub 2010 Dec 8. Nat Rev Mol Cell Biol. 2011. PMID: 21139638 Review.
-
OEP61 is a chaperone receptor at the plastid outer envelope.Biochem J. 2011 Aug 15;438(1):143-53. doi: 10.1042/BJ20110448. Biochem J. 2011. PMID: 21612577 Free PMC article.
References
-
- Abe, Y., T. Shodai, T. Muto, K. Mihara, H. Torii, S. Nishikawa, T. Endo, and D. Kohda. 2000. Structural basis of presequence recognition by the mitochondrial protein import receptor Tom20. Cell. 100:551–560. - PubMed
-
- Artigues, A., A. Iriarte, and M. Martinez-Carrion. 2002. Binding to chaperones allows import of a purified mitochondrial precursor into mitochondria. J. Biol. Chem. 277:25047–25055. - PubMed
-
- Bell, D.R., and A. Poland. 2000. Binding of aryl hydrocarbon receptor (AhR) to AhR-interacting protein. The role of hsp90. J. Biol. Chem. 275:36407–36414. - PubMed
-
- Bose, S., T. Weikl, H. Bugl, and J. Buchner. 1996. Chaperone function of Hsp90-associated proteins. Science. 274:1715–1717. - PubMed
-
- Carver, L.A., and C.A. Bradfield. 1997. Ligand-dependent interaction of the aryl hydrocarbon receptor with a novel immunophilin homolog in vivo. J. Biol. Chem. 272:11452–11456. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

