Two distinct size classes of immature and mature subviral particles from tick-borne encephalitis virus
- PMID: 14557621
- PMCID: PMC229348
- DOI: 10.1128/jvi.77.21.11357-11366.2003
Two distinct size classes of immature and mature subviral particles from tick-borne encephalitis virus
Abstract
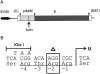
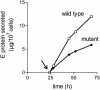
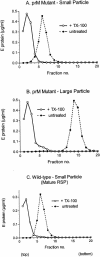
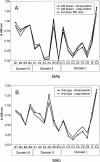
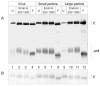
Flaviviruses assemble in the endoplasmic reticulum by a mechanism that appears to be driven by lateral interactions between heterodimers of the envelope glycoproteins E and prM. Immature intracellular virus particles are then transported through the secretory pathway and converted to their mature form by cleavage of the prM protein by the cellular protease furin. Earlier studies showed that when the prM and E proteins of tick-borne encephalitis virus are expressed together in mammalian cells, they assemble into membrane-containing, icosahedrally symmetrical recombinant subviral particles (RSPs), which are smaller than whole virions but retain functional properties and undergo cleavage maturation, yielding a mature form in which the E proteins are arranged in a regular T = 1 icosahedral lattice. In this study, we generated immature subviral particles by mutation of the furin recognition site in prM. The mutation resulted in the secretion of two distinct size classes of particles that could be separated by sucrose gradient centrifugation. Electron microscopy showed that the smaller particles were approximately the same size as the previously described mature RSPs, whereas the larger particles were approximately the same size as the virus. Particles of the larger size class were also detected with a wild-type construct that allowed prM cleavage, although in this case the smaller size class was far more prevalent. Subtle differences in endoglycosidase sensitivity patterns suggested that, in contrast to the small particles, the E glycoproteins in the large subviral particles and whole virions might be in nonequivalent structural environments during intracellular transport, with a portion of them inaccessible to cellular glycan processing enzymes. These proteins thus appear to have the intrinsic ability to form alternative assembly products that could provide important clues about the role of lateral envelope protein interactions in flavivirus assembly.
Figures

Similar articles
-
Intracellular assembly and secretion of recombinant subviral particles from tick-borne encephalitis virus.J Virol. 2003 Apr;77(7):4370-82. doi: 10.1128/jvi.77.7.4370-4382.2003. J Virol. 2003. PMID: 12634393 Free PMC article.
-
Recombinant subviral particles from tick-borne encephalitis virus are fusogenic and provide a model system for studying flavivirus envelope glycoprotein functions.J Virol. 1996 Jul;70(7):4549-57. doi: 10.1128/JVI.70.7.4549-4557.1996. J Virol. 1996. PMID: 8676481 Free PMC article.
-
Cleavage of protein prM is necessary for infection of BHK-21 cells by tick-borne encephalitis virus.J Gen Virol. 2003 Jan;84(Pt 1):183-191. doi: 10.1099/vir.0.18723-0. J Gen Virol. 2003. PMID: 12533715
-
Degrees of maturity: the complex structure and biology of flaviviruses.Curr Opin Virol. 2012 Apr;2(2):168-75. doi: 10.1016/j.coviro.2012.02.011. Epub 2012 Mar 23. Curr Opin Virol. 2012. PMID: 22445964 Free PMC article. Review.
-
Post-translational regulation and modifications of flavivirus structural proteins.J Gen Virol. 2015 Jul;96(Pt 7):1551-69. doi: 10.1099/vir.0.000097. Epub 2015 Feb 23. J Gen Virol. 2015. PMID: 25711963 Review.
Cited by
-
Functional requirements of the yellow fever virus capsid protein.J Virol. 2007 Jun;81(12):6471-81. doi: 10.1128/JVI.02120-06. J Virol. 2007. PMID: 17526891 Free PMC article.
-
Investigating Tick-borne Flaviviral-like Particles as a Delivery System for Gene Therapy.Curr Ther Res Clin Exp. 2017 Oct 16;88:8-17. doi: 10.1016/j.curtheres.2017.10.003. eCollection 2018. Curr Ther Res Clin Exp. 2017. PMID: 30093925 Free PMC article.
-
Genome cyclization as strategy for flavivirus RNA replication.Virus Res. 2009 Feb;139(2):230-9. doi: 10.1016/j.virusres.2008.07.016. Epub 2008 Sep 9. Virus Res. 2009. PMID: 18703097 Free PMC article. Review.
-
The C-terminal helical domain of dengue virus precursor membrane protein is involved in virus assembly and entry.Virology. 2011 Feb 5;410(1):170-80. doi: 10.1016/j.virol.2010.11.006. Epub 2010 Dec 3. Virology. 2011. PMID: 21129763 Free PMC article.
-
Identification of a critical role for ZIKV capsid α3 in virus assembly and its genetic interaction with M protein.PLoS Negl Trop Dis. 2024 Jan 2;18(1):e0011873. doi: 10.1371/journal.pntd.0011873. eCollection 2024 Jan. PLoS Negl Trop Dis. 2024. PMID: 38166143 Free PMC article.
References
-
- Allison, S. L., C. W. Mandl, C. Kunz, and F. X. Heinz. 1994. Expression of cloned envelope protein genes from the flavivirus tick-borne encephalitis virus in mammalian cells and random mutagenesis by PCR. Virus Genes 8:187-198. - PubMed
-
- Corver, J., A. Ortiz, S. L. Allison, J. Schalich, F. X. Heinz, and J. Wilschut. 2000. Membrane fusion activity of tick-borne encephalitis virus and recombinant subviral particles in a liposomal model system. Virology 269:37-46. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

