Myosin-Va binds to and mechanochemically couples microtubules to actin filaments
- PMID: 14565972
- PMCID: PMC307536
- DOI: 10.1091/mbc.e03-07-0504
Myosin-Va binds to and mechanochemically couples microtubules to actin filaments
Abstract
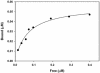
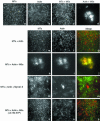
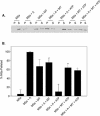
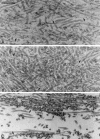
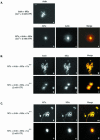
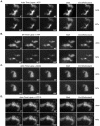
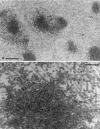
Myosin-Va was identified as a microtubule binding protein by cosedimentation analysis in the presence of microtubules. Native myosin-Va purified from chick brain, as well as the expressed globular tail domain of this myosin, but not head domain bound to microtubule-associated protein-free microtubules. Binding of myosin-Va to microtubules was saturable and of moderately high affinity (approximately 1:24 Myosin-Va:tubulin; Kd = 70 nM). Myosin-Va may bind to microtubules via its tail domain because microtubule-bound myosin-Va retained the ability to bind actin filaments resulting in the formation of cross-linked gels of microtubules and actin, as assessed by fluorescence and electron microscopy. In low Ca2+, ATP addition induced dissolution of these gels, but not release of myosin-Va from MTs. However, in 10 microM Ca2+, ATP addition resulted in the contraction of the gels into aster-like arrays. These results demonstrate that myosin-Va is a microtubule binding protein that cross-links and mechanochemically couples microtubules to actin filaments.
Figures
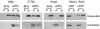

References
-
- Burns, R.G., and Islam, K. (1984). Stoichiometry of microtubule-associated protein (MAP2):tubulin and the localisation of the phosphorylation and cysteine residues along the MAP2 primary sequence. Eur. J. Biochem. 141, 599–608. - PubMed
-
- Cheney, R.E. (1998). Purification and assay of myosin V. Methods Enzymol. 298, 3–18. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

