Lipid-dependent bidirectional traffic of apolipoprotein B in polarized enterocytes
- PMID: 14565984
- PMCID: PMC307534
- DOI: 10.1091/mbc.e03-04-0215
Lipid-dependent bidirectional traffic of apolipoprotein B in polarized enterocytes
Abstract
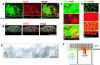
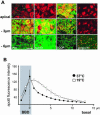
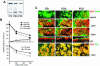
Enterocytes are highly polarized cells that transfer nutrients across the intestinal epithelium from the apical to the basolateral pole. Apolipoprotein B (apoB) is a secretory protein that plays a key role in the transepithelial transport of dietary fatty acids as triacylglycerol. The evaluation of the control of apoB traffic by lipids is therefore of particular interest. To get a dynamic insight into this process, we used the enterocytic Caco-2 cells cultured on microporous filters, a system in which the apical and basal compartments can be delimited. Combining biochemical and morphological approaches, our results showed that, besides their role in protection from degradation, lipids control the intracellular traffic of apoB in enterocytes. A supply of fatty acids and cholesterol is sufficient for the export of apoB from the endoplasmic reticulum and its post-Golgi traffic up to the apical brush-border domain, where it remains until an apical supply of complex lipid micelles signals its chase down to the basolateral secretory domain. This downward traffic of apoB involves a microtubule-dependent process. Our results demonstrate an enterocyte-specific bidirectional process for the lipid-dependent traffic of a secretory protein.
Figures
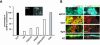




References
-
- Abumrad, N., Coburn, C., and Ibrahimi, A. (1999). Membrane proteins implicated in long-chain fatty acid uptake by mammalian cells: CD36, FATP and FABPm. Biochim. Biophys. Acta 1441, 4–13. - PubMed
-
- Boren, J., Rustaeus, S., and Olofsson, S.O. (1994). Studies on the assembly of apolipoprotein B-100- and B-48-containing very low density lipoproteins in McA-RH7777 cells. J. Biol. Chem. 269, 25879–25888. - PubMed
-
- Bostrom, K., Boren, J., Wettesten, M., Sjoberg, A., Bondjers, G., Wiklund, O., Carlsson, P., and Olofsson, S.O. (1988). Studies on the assembly of apo B-100-containing lipoproteins in HepG2 cells. J. Biol. Chem. 263, 4434–4442. - PubMed
-
- Carpentier, A., Taghibiglou, C., Leung, N., Szeto, L., Van Iderstine, S.C., Uffelman, K.D., Buckingham, R., Adeli, K., and Lewis, G.F. (2002). Ameliorated hepatic insulin resistance is associated with normalization of microsomal triglyceride transfer protein expression and reduction in very low density lipoprotein assembly and secretion in the fructose-fed hamster. J. Biol. Chem. 277, 28795–28802. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

