Measuring Solution Viscosity and its Effect on Enzyme Activity
- PMID: 14569610
- PMCID: PMC154660
- DOI: 10.1251/bpo52
Measuring Solution Viscosity and its Effect on Enzyme Activity
Abstract
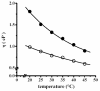
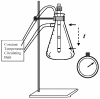
In proteins, some processes require conformational changes involving structural domain diffusion. Among these processes are protein folding, unfolding and enzyme catalysis. During catalysis some enzymes undergo large conformational changes as they progress through the catalytic cycle. According to Kramers theory, solvent viscosity results in friction against proteins in solution, and this should result in decreased motion, inhibiting catalysis in motile enzymes. Solution viscosity was increased by adding increasing concentrations of glycerol, sucrose and trehalose, resulting in a decrease in the reaction rate of the H(+)-ATPase from the plasma membrane of Kluyveromyces lactis. A direct correlation was found between viscosity (eta) and the inhibition of the maximum rate of catalysis (V(max)). The protocol used to measure viscosity by means of a falling ball type viscometer is described, together with the determination of enzyme kinetics and the application of Kramers' equation to evaluate the effect of viscosity on the rate of ATP hydrolysis by the H(+)-ATPase.
Figures


References
-
- Kramers HA. Brownian motion in a field of force and the diffusion model of chemical reactions. Physica. 1940;7:284–304.
-
- Jacob M, Geeves M, Holterman G, Schmid FX. Diffusional crossing in a two-state protein folding reaction. Nature Struc Biol. 1999;6:923–926. - PubMed
-
- Demchenko AP, Ruskyn OI, Saburova EA. Kinetics of the lactate dehydrogenase reaction in high-viscosity media. Biochim Biophys Acta. 1989;998:196–203. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

