PA-457: a potent HIV inhibitor that disrupts core condensation by targeting a late step in Gag processing
- PMID: 14573704
- PMCID: PMC263852
- DOI: 10.1073/pnas.2234683100
PA-457: a potent HIV inhibitor that disrupts core condensation by targeting a late step in Gag processing
Abstract
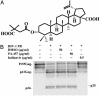
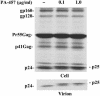
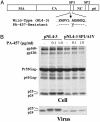
New HIV therapies are urgently needed to address the growing problem of drug resistance. In this article, we characterize the anti-HIV drug candidate 3-O-(3',3'-dimethylsuccinyl) betulinic acid (PA-457). We show that PA-457 potently inhibits replication of both WT and drug-resistant HIV-1 isolates and demonstrate that the compound acts by disrupting a late step in Gag processing involving conversion of the capsid precursor (p25) to mature capsid protein (p24). We find that virions from PA-457-treated cultures are noninfectious and exhibit an aberrant particle morphology characterized by a spherical, acentric core and a crescent-shaped, electron-dense shell lying just inside the viral membrane. To identify the determinants of compound activity we selected for PA-457-resistant virus in vitro. Consistent with the effect on Gag processing, we found that mutations conferring resistance to PA-457 map to the p25 to p24 cleavage site. PA-457 represents a unique class of anti-HIV compounds termed maturation inhibitors that exploit a previously unidentified viral target, providing additional opportunities for HIV drug discovery.
Figures


References
-
- LaBonte, J., Lebbos, J. & Kirkpatrick, P. (2003) Nat. Rev. Drug Discov. 2, 345-346. - PubMed
-
- Grant, R. M., Hecht, F. M., Warmerdam, M., Liu, L., Liegler, T., Petropoulos, C. J., Hellmann, N. S., Chesney, M., Busch, M. P., Kahn, J. O., et al. (2002) J. Am. Med. Assoc. 288, 181-188. - PubMed
-
- Little, S. J., Holte, S., Routy, J. P., Daar, E. S., Markowitz, M., Collier, A. C., Koup, R. A., Mellors, J. W., Connick, E., Conway, B., et al. (2002) N. Engl. J. Med. 347, 385-394. - PubMed
-
- Swanstrom, R. & Wills, J. W. (1997) in Retroviruses, eds. Weiss, R., Teich, N., Varmus, H. & Coffin, J. M. (Cold Spring Harbor Lab. Press, Plainview, NY), pp. 263-334.
-
- Freed, E. O. (1998) Virology 251, 1-15. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

