Role of tissue factor and protease-activated receptors in a mouse model of endotoxemia
- PMID: 14576054
- PMCID: PMC2860856
- DOI: 10.1182/blood-2003-09-3051
Role of tissue factor and protease-activated receptors in a mouse model of endotoxemia
Abstract
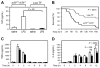
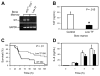
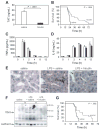
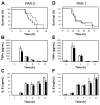
Sepsis is associated with a systemic activation of coagulation and an excessive inflammatory response. Anticoagulants have been shown to inhibit both coagulation and inflammation in sepsis. In this study, we used both genetic and pharmacologic approaches to analyze the role of tissue factor and protease-activated receptors in coagulation and inflammation in a mouse endotoxemia model. We used mice expressing low levels of the procoagulant molecule, tissue factor (TF), to analyze the effects of TF deficiency either in all tissues or selectively in hematopoietic cells. Low TF mice had reduced coagulation, inflammation, and mortality compared with control mice. Similarly, a deficiency of TF expression by hematopoietic cells reduced lipopolysaccharide (LPS)-induced coagulation, inflammation, and mortality. Inhibition of the down-stream coagulation protease, thrombin, reduced fibrin deposition and prolonged survival without affecting inflammation. Deficiency of either protease activated receptor-1 (PAR-1) or protease activated receptor-2 (PAR-2) alone did not affect inflammation or survival. However, a combination of thrombin inhibition and PAR-2 deficiency reduced inflammation and mortality. These data demonstrate that hematopoietic cells are the major pathologic site of TF expression during endotoxemia and suggest that multiple protease-activated receptors mediate crosstalk between coagulation and inflammation.
Figures

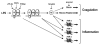
References
-
- Parrillo JE. Pathogenetic mechanisms of septic shock. N Engl J Med. 1993;328:1471–1477. - PubMed
-
- Riedemann NC, Guo RF, Ward PA. Novel strategies for the treatment of sepsis. Nat Med. 2003;9:517–524. - PubMed
-
- Taylor FB, Jr, Emerson TE, Jr, Jordan R, Chang AK, Blick KE. Antithrombin-III prevents the lethal effects of Escherichia coli infusion in baboons. Circ Shock. 1998;26:227–235. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

