Na/Ca exchange and Na/K-ATPase function are equally concentrated in transverse tubules of rat ventricular myocytes
- PMID: 14581240
- PMCID: PMC1303616
- DOI: 10.1016/S0006-3495(03)74758-4
Na/Ca exchange and Na/K-ATPase function are equally concentrated in transverse tubules of rat ventricular myocytes
Abstract
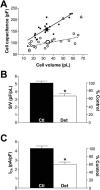
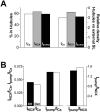
Formamide-induced detubulation of rat ventricular myocytes was used to investigate the functional distribution of the Na/Ca exchanger (NCX) and Na/K-ATPase between the t-tubules and external sarcolemma. Detubulation resulted in a 32% decrease in cell capacitance, whereas cell volume was unchanged. Thus, the surface-to-volume ratio was used to assess the success of detubulation. NCX current (I(NCX)) and Na/K pump current (I(pump)) were recorded using whole-cell patch clamp, as Cd-sensitive and K-activated currents, respectively. Both inward and outward I(NCX) density was significantly reduced by approximately 40% in detubulated cells. I(NCX) density at 0 mV decreased from 0.19 +/- 0.03 to 0.10 +/- 0.03 pA/pF upon detubulation. I(pump) density was also lower in detubulated myocytes over the range of voltages (-50 to +100 mV) and internal [Na] ([Na](i)) investigated (7-22 mM). At [Na](i) = 10 mM and -20 mV, I(pump) density was reduced by 39% in detubulated myocytes (0.28 +/- 0.02 vs. 0.17 +/- 0.03 pA/pF), but the apparent K(m) for [Na](i) was unchanged (16.9 +/- 0.4 vs. 17.0 +/- 0.3 mM). These results indicate that although thet-tubules represent only approximately 32% of the total sarcolemma, they contribute approximately 60% to the total I(NCX) and I(pump). Thus, the functional density of NCX and Na/K pump in the t-tubules is 3-3.5-fold higher than in the external sarcolemma.
Figures

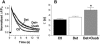


References
-
- Bers, D. M. 2001. Excitation-contraction coupling and cardiac contractile force. Kluwer Academic Publishers, Dordrecht, The Netherlands.
-
- Bers, D. M., W. H. Barry, and S. Despa. 2003. Intracellular Na+ regulation in cardiac myocytes. Cardiovasc. Res. 57:897–912. - PubMed
-
- Blanco, G., and R. W. Mercer. 1998. Isozymes of the Na-K-ATPase: heterogeneity in structure, diversity in function. Am. J. Physiol. 275:F633–F650. - PubMed
-
- Boyett, M., J. E. Frampton, and M. S. Kirby. 1991. The length, width and volume of isolated rat and ferret ventricular myocytes during twitch contractions and change in osmotic strength. Exp. Physiol. 76:259–270. - PubMed
-
- Brette F., K. Komukai, and C. H. Orchard. 2002. Validation of formamide as a detubulation agent in isolated rat cardiac cells. Am. J. Physiol. 283:H1720–H1728. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

