Dominant role of smooth muscle L-type calcium channel Cav1.2 for blood pressure regulation
- PMID: 14609949
- PMCID: PMC275441
- DOI: 10.1093/emboj/cdg583
Dominant role of smooth muscle L-type calcium channel Cav1.2 for blood pressure regulation
Abstract
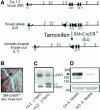
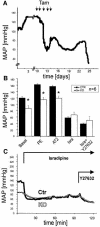
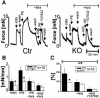
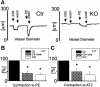
Blood pressure is regulated by a number of key molecules involving G-protein-coupled receptors, ion channels and monomeric small G-proteins. The relative contribution of these different signaling pathways to blood pressure regulation remains to be determined. Tamoxifen-induced, smooth muscle-specific inactivation of the L-type Cav1.2 Ca2+ channel gene in mice (SMAKO) reduced mean arterial blood pressure (MAP) in awake, freely moving animals from 120 +/- 4.5 to 87 +/- 8 mmHg. Phenylephrine (PE)- and angiotensin 2 (AT2)-induced MAP increases were blunted in SMAKO mice, whereas the Rho-kinase inhibitor Y-27632 reduced MAP to the same extent in control and SMAKO mice. Depolarization-induced contraction was abolished in tibialis arteries of SMAKO mice, and development of myogenic tone in response to intravascular pressure (Bayliss effect) was absent. Hind limb perfusion experiments suggested that 50% of the PE-induced resistance is due to calcium influx through the Cav1.2 channel. These results show that Cav1.2 calcium channels are key players in the hormonal regulation of blood pressure and development of myogenic tone.
Figures



References
-
- Bolz S.S., Vogel,L., Sollinger,D., Derwand,R., Boer,C., Pitson,S.M., Spiegel,S. and Pohl,U. (2003) Sphingosine kinase modulates microvascular tone and myogenic responses through activation of RhoA/Rho kinase. Circulation, 108, 342–347. - PubMed
-
- Brandes R.P., Schmitz-Winnenthal,F.H., Feletou,M., Godecke,A., Huang,P.L., Vanhoutte,P.M., Fleming,I. and Busse,R. (2000) An endothelium-derived hyperpolarizing factor distinct from NO and prostacyclin is a major endothelium-dependent vasodilator in resistance vessels of wild-type and endothelial NO synthase knockout mice. Proc. Natl Acad. Sci. USA, 97, 9747–9752. - PMC - PubMed
-
- Brayden J.E. and Nelson,M.T. (1992) Regulation of arterial tone by activation of calcium-dependent potassium channels. Science, 256, 532–535. - PubMed
-
- Davis M.J. and Hill,M.A. (1999) Signaling mechanisms underlying the vascular myogenic response. Physiol. Rev., 79, 387–423. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

