Cell-type-specific activation of PAK2 by transforming growth factor beta independent of Smad2 and Smad3
- PMID: 14612425
- PMCID: PMC262664
- DOI: 10.1128/MCB.23.23.8878-8889.2003
Cell-type-specific activation of PAK2 by transforming growth factor beta independent of Smad2 and Smad3
Abstract
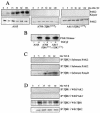
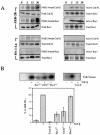
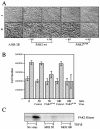
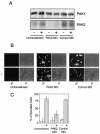
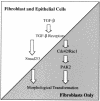
Transforming growth factor beta (TGF-beta) causes growth arrest in epithelial cells and proliferation and morphological transformation in fibroblasts. Despite the ability of TGF-beta to induce various cellular phenotypes, few discernible differences in TGF-beta signaling between cell types have been reported, with the only well-characterized pathway (the Smad cascade) seemingly under identical control. We determined that TGF-beta receptor signaling activates the STE20 homolog PAK2 in mammalian cells. PAK2 activation occurs in fibroblast but not epithelial cell cultures and is independent of Smad2 and/or Smad3. Furthermore, we show that TGF-beta-stimulated PAK2 activity is regulated by Rac1 and Cdc42 and dominant negative PAK2 or morpholino antisense oligonucleotides to PAK2 prevent the morphological alteration observed following TGF-beta addition. Thus, PAK2 represents a novel Smad-independent pathway that differentiates TGF-beta signaling in fibroblast (growth-stimulated) and epithelial cell (growth-inhibited) cultures.
Figures



Similar articles
-
Transforming growth factor beta signaling via Ras in mesenchymal cells requires p21-activated kinase 2 for extracellular signal-regulated kinase-dependent transcriptional responses.Cancer Res. 2007 Apr 15;67(8):3673-82. doi: 10.1158/0008-5472.CAN-06-3211. Cancer Res. 2007. PMID: 17440079
-
Transforming growth factor beta activation of c-Abl is independent of receptor internalization and regulated by phosphatidylinositol 3-kinase and PAK2 in mesenchymal cultures.J Biol Chem. 2006 Sep 22;281(38):27846-54. doi: 10.1074/jbc.M603721200. Epub 2006 Jul 24. J Biol Chem. 2006. PMID: 16867995
-
Imatinib mesylate blocks a non-Smad TGF-beta pathway and reduces renal fibrogenesis in vivo.FASEB J. 2005 Jan;19(1):1-11. doi: 10.1096/fj.04-2370com. FASEB J. 2005. PMID: 15629889
-
Transforming growth factor-beta activation of phosphatidylinositol 3-kinase is independent of Smad2 and Smad3 and regulates fibroblast responses via p21-activated kinase-2.Cancer Res. 2005 Nov 15;65(22):10431-40. doi: 10.1158/0008-5472.CAN-05-1522. Cancer Res. 2005. PMID: 16288034
-
The transcriptional co-activator P/CAF potentiates TGF-beta/Smad signaling.Nucleic Acids Res. 2000 Nov 1;28(21):4291-8. doi: 10.1093/nar/28.21.4291. Nucleic Acids Res. 2000. PMID: 11058129 Free PMC article.
Cited by
-
Profibrotic up-regulation of glucose transporter 1 by TGF-β involves activation of MEK and mammalian target of rapamycin complex 2 pathways.FASEB J. 2016 Nov;30(11):3733-3744. doi: 10.1096/fj.201600428R. Epub 2016 Aug 1. FASEB J. 2016. PMID: 27480571 Free PMC article.
-
TGF-β: The missing link in obesity-associated airway diseases?Curr Res Pharmacol Drug Discov. 2021 Feb 5;2:100016. doi: 10.1016/j.crphar.2021.100016. eCollection 2021. Curr Res Pharmacol Drug Discov. 2021. PMID: 34909651 Free PMC article. Review.
-
Pak2-mediated phosphorylation promotes RORγt ubiquitination and inhibits colonic inflammation.Cell Rep. 2022 Sep 13;40(11):111345. doi: 10.1016/j.celrep.2022.111345. Cell Rep. 2022. PMID: 36103814 Free PMC article.
-
Specificity, versatility, and control of TGF-β family signaling.Sci Signal. 2019 Feb 26;12(570):eaav5183. doi: 10.1126/scisignal.aav5183. Sci Signal. 2019. PMID: 30808818 Free PMC article. Review.
-
Non-Smad Signaling Pathways of the TGF-β Family.Cold Spring Harb Perspect Biol. 2017 Feb 1;9(2):a022129. doi: 10.1101/cshperspect.a022129. Cold Spring Harb Perspect Biol. 2017. PMID: 27864313 Free PMC article. Review.
References
-
- Anders, R. A., J. E. J. Doré, S. A. Arline, N. Garamszegi, and E. B. Leof. 1998. Differential requirements for type I and type II TGFβ receptor kinase activity in ligand-mediated receptor endocytosis. J. Biol. Chem. 273:23118-23125. - PubMed
-
- Anders, R. A., and E. B. Leof. 1996. Chimeric granulocyte/macrophage colony-stimulating factor/transforming growth factor-β (TGF-β) receptors define a model system for investigating the role of homomeric and heteromeric receptors in TGF-β signaling. J. Biol. Chem. 271:21758-21766. - PubMed
-
- Bagrodia, S., and R. A. Cerione. 1999. PAK to the future. Trends Cell Biol. 9:350-355. - PubMed
-
- Bissell, D. M. 2001. Chronic liver injury, TGF-β, and cancer. Exp. Mol. Med. 33:179-190. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
