Coadministration of voriconazole and phenytoin: pharmacokinetic interaction, safety, and toleration
- PMID: 14616412
- PMCID: PMC1884312
- DOI: 10.1046/j.1365-2125.2003.01997.x
Coadministration of voriconazole and phenytoin: pharmacokinetic interaction, safety, and toleration
Abstract
Aims: Voriconazole is a new triazole antifungal agent, and is metabolized by the cytochrome P450 isoenzymes CYP2C9, CYP2C19, and, to a lesser extent, by CYP3A4. Phenytoin is an inducer of CYP3A4 activity, and a substrate and inducer of CYP2C9 and CYP2C19. The present studies investigated the pharmacokinetic interactions of voriconazole and phenytoin when coadministered.
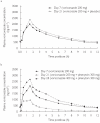
Methods: Two placebo-controlled parallel-group studies were conducted in healthy male volunteers. Study A was an open-label study and investigated the effect of phenytoin (300 mg once daily) on the steady-state pharmacokinetics of voriconazole (200 mg and 400 mg twice daily). Study B was a double-blind randomized study to investigate the effects of voriconazole (400 mg twice daily) on the steady-state pharmacokinetics of phenytoin (300 mg once daily). Cmax and AUCtau were compared at days 7, 21, and 28 (Study A), and at days 7 and 17 (Study B). All adverse events were recorded.
Results: Study A: 21 subjects were evaluable (10 voriconazole + phenytoin, 11 voriconazole + placebo). For subjects receiving voriconazole (200 mg twice daily) plus phenytoin, the day 21/day 7 ratios for voriconazole Cmax and AUCtau were 60.7%[90% confidence interval (CI) 50.1, 73.6] and 35.9% (90% CI 29.7, 43.3), respectively. Adjusted for voriconazole + placebo, the ratios between the means were 50.7% (90% CI 38.8, 66.1) and 30.6% (90% CI 23.5, 39.7), respectively. When the dose of voriconazole was increased to 400 mg twice daily, the day 28/day 7 ratios for voriconazole Cmax and AUCtau were 134% (90% CI 89.2, 200) and 139% (90% CI 97.3, 199), respectively. Study B: 15 subjects were evaluable for pharmacokinetic assessments (six phenytoin + voriconazole, nine phenytoin + placebo). The ratios between the means for phenytoin + voriconazole/phenytoin + placebo on day 17 vs. day 7 were: phenytoin Cmax 167% (90% CI 144, 193) and phenytoin AUCtau 181% (90% CI 156, 210). All treatments were well tolerated: most adverse events were mild/moderate and transient.
Conclusions: Repeat dose administration of phenytoin decreased the mean steady-state Cmax and AUCtau of voriconazole by approximately 50% and 70%, respectively. Increasing the dose of voriconazole from 200 mg to 400 mg b.d. compensated for this effect. Repeat dose administration of 400 mg b.d. voriconazole increased the mean steady-state Cmax and AUCtau of phenytoin by approximately 70% and 80%, respectively. It is therefore recommended that plasma phenytoin concentrations are monitored and the dose adjusted as appropriate when phenytoin is coadministered with voriconazole.
Figures

Similar articles
-
No clinically significant pharmacokinetic interactions between voriconazole and indinavir in healthy volunteers.Br J Clin Pharmacol. 2003 Dec;56 Suppl 1(Suppl 1):62-8. doi: 10.1046/j.1365-2125.2003.02001.x. Br J Clin Pharmacol. 2003. PMID: 14616416 Free PMC article. Clinical Trial.
-
Effect of omeprazole on the steady-state pharmacokinetics of voriconazole.Br J Clin Pharmacol. 2003 Dec;56 Suppl 1(Suppl 1):56-61. doi: 10.1046/j.1365-2125.2003.02000.x. Br J Clin Pharmacol. 2003. PMID: 14616415 Free PMC article. Clinical Trial.
-
No clinically significant effect of erythromycin or azithromycin on the pharmacokinetics of voriconazole in healthy male volunteers.Br J Clin Pharmacol. 2003 Dec;56 Suppl 1(Suppl 1):30-6. doi: 10.1046/j.1365-2125.2003.01996.x. Br J Clin Pharmacol. 2003. PMID: 14616411 Free PMC article. Clinical Trial.
-
Pharmacokinetic/pharmacodynamic profile of voriconazole.Clin Pharmacokinet. 2006;45(7):649-63. doi: 10.2165/00003088-200645070-00002. Clin Pharmacokinet. 2006. PMID: 16802848 Review.
-
Pharmacogenomics of the triazole antifungal agent voriconazole.Pharmacogenomics. 2011 Jun;12(6):861-72. doi: 10.2217/pgs.11.18. Pharmacogenomics. 2011. PMID: 21692616 Review.
Cited by
-
Clinical Pharmacokinetics of Second-Generation Triazoles for the Treatment of Invasive Aspergillosis and Candidiasis.Eur J Drug Metab Pharmacokinet. 2019 Apr;44(2):139-157. doi: 10.1007/s13318-018-0513-7. Eur J Drug Metab Pharmacokinet. 2019. PMID: 30284178 Review.
-
Expansion of a PBPK model to predict disposition in pregnant women of drugs cleared via multiple CYP enzymes, including CYP2B6, CYP2C9 and CYP2C19.Br J Clin Pharmacol. 2014 Mar;77(3):554-70. doi: 10.1111/bcp.12207. Br J Clin Pharmacol. 2014. PMID: 23834474 Free PMC article.
-
Clinical Pharmacogenetics Implementation Consortium (CPIC) Guideline for CYP2C9 and HLA-B Genotypes and Phenytoin Dosing: 2020 Update.Clin Pharmacol Ther. 2021 Feb;109(2):302-309. doi: 10.1002/cpt.2008. Epub 2020 Sep 6. Clin Pharmacol Ther. 2021. PMID: 32779747 Free PMC article.
-
The enzymatic basis of drug-drug interactions with systemic triazole antifungals.Clin Pharmacokinet. 2008;47(12):779-92. doi: 10.2165/0003088-200847120-00003. Clin Pharmacokinet. 2008. PMID: 19026034 Review.
-
Clinical pharmacogenetics implementation consortium guidelines for CYP2C9 and HLA-B genotypes and phenytoin dosing.Clin Pharmacol Ther. 2014 Nov;96(5):542-8. doi: 10.1038/clpt.2014.159. Epub 2014 Aug 6. Clin Pharmacol Ther. 2014. PMID: 25099164 Free PMC article.
References
-
- Cuenca-Estrella M, Rodriguez-Tudela JL, Mellado E, Martinez-Suarez JV, Monzon A. Comparison of the in vitro activity of voriconazole (UK-109,496), itraconazole and amphotericin B against clinical isolates of Aspergillus fumigatus. J Antimicrob Chemother. 1998;42:531–533. - PubMed
-
- Chandrasekar PH, Manavathu E. Voriconazole: a second-generation triazole. Drugs Today. 2001;37:135–148. - PubMed
-
- Torre-Cisneros J, Gonzalez-Ruiz A, Hodges MR, Lutsar I. Program and Abstracts of the 38th Annual Meeting of the Infectious Diseases Society of America. New Orleans, LA: September 2000. Voriconazole (VORI) for the treatment of S. apiospermum and S. prolificans infection. Abstract 305, September.
-
- Perfect J, Gonzalez-Ruiz A, Lutsar I. Program and Abstracts of the 38th Annual Meeting of the Infectious Diseases Society of America. New Orleans, LA: September 2000. Voriconazole (VORI) for the treatment of resistant and rare fungal pathogens. Abstract 303, September.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

