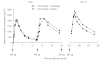Effect of omeprazole on the steady-state pharmacokinetics of voriconazole
- PMID: 14616415
- PMCID: PMC1884317
- DOI: 10.1046/j.1365-2125.2003.02000.x
Effect of omeprazole on the steady-state pharmacokinetics of voriconazole
Abstract
Aims: Voriconazole, a novel triazole antifungal agent, is metabolized by the cytochrome P450 isoenzymes CYP2C19, CYP2C9, and to a lesser extent by CYP3A4. Omeprazole, a proton pump inhibitor used widely for the treatment of gastric and duodenal ulcers, is predominantly metabolized by CYP2C19 and CYP3A4. The aim of this study was to determine the effects of omeprazole on the steady-state pharmacokinetics of voriconazole. A secondary objective was to characterize the pharmacokinetic profile of an oral loading dose regimen of 400 mg twice-daily voriconazole on day 1.
Methods: This was an open, randomized, placebo-controlled, two-way crossover study of 18 healthy male volunteers. Subjects received oral voriconazole (400 mg twice daily on day 1 followed by 200 mg twice daily on days 2-9 and a single 200-mg dose on day 10) with either omeprazole (40 mg once daily) or matched placebo for 10 days. There was a minimum 7-day washout between treatment periods.
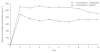
Results: Mean Cmax and AUCtau of voriconazole were increased by 15%[90% confidence interval (CI) 5, 25] and 41% (90% CI 29, 55), respectively, with no effect on tmax during coadministration of omeprazole. Visual inspection of predose plasma concentrations (Cmin) indicated that steady-state plasma concentrations were achieved following the second loading dose. One subject withdrew from the study during the voriconazole + omeprazole treatment period because of treatment-related abnormal liver function test values. All other treatment-related adverse events resolved without intervention.
Conclusions: Omeprazole had no clinically relevant effect on voriconazole exposure, suggesting that no voriconazole dosage adjustment is necessary for patients in whom omeprazole therapy is initiated. Administration of a 400-mg twice-daily oral loading dose regimen on day 1 resulted in steady-state plasma levels of voriconazole being achieved following the second loading dose.
Figures
References
-
- Cuenca-Estrella M, Rodriguez-Tudela JL, Mellado E, Martinez-Suarez JV, Monzon A. Comparison of the in vitro activity of voriconazole (UK-109,496), itraconazole and amphotericin B against clinical isolates of Aspergillus fumigatus. J Antimicrob Chemother. 1998;42:531–533. - PubMed
-
- Chandrasekar PH, Manavathu E. Voriconazole: a second-generation triazole. Drugs Today. 2001;37:135–148. - PubMed
-
- Torre-Cisneros J, Gonzalez-Ruiz A, Hodges MR, Lutsar I. Program and Abstracts of the 38th Annual Meeting of the Infectious Diseases Society of America. New Orleans, LA: 2000. Voriconazole (VORI) for the treatment of S. apiospermum and S. prolificans infection. Abstract 305, September.
-
- Perfect J, Gonzalez-Ruiz A, Lutsar I. Program and Abstracts of the 38th Annual Meeting of the Infectious Diseases Society of America. New Orleans, LA: 2000. Voriconazole (VORI) for the treatment of resistant and rare fungal pathogens. Abstract 303, September.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources