Identification and preliminary characterization of a 75-kDa hemin- and hemoglobin-binding outer membrane protein of Actinobacillus pleuropneumoniae serotype 1
- PMID: 14620863
- PMCID: PMC280711
Identification and preliminary characterization of a 75-kDa hemin- and hemoglobin-binding outer membrane protein of Actinobacillus pleuropneumoniae serotype 1
Abstract
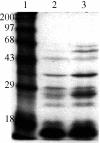
The reference strains representing serotypes 1 to 12 of Actinobacillus pleuropneumoniae biotype 1 were examined for their ability to utilize porcine hemoglobin (Hb) or porcine hemin (Hm) as iron sources for growth. In a growth promotion assay, all of the reference strains were able to use porcine Hb, and all strains except 2 were able to use porcine Hm. Using a preliminary characterization procedure with Hm- or Hb-agarose, Hm- and Hb-binding outer membrane proteins (OMPs) of approximately 75 kDa were isolated from A. pleuropneumoniae serotype 1 strain 4074 grown under iron-restricted conditions. Matrix-assisted laser desorption ionization/time-of-flight (MALDI-TOF) analysis revealed a number of common tryptic peptides between the Hb-agarose- and Hm-agarose-purified 75 kDa OMPs, strongly suggesting that these peptides originate from the same protein. A database search of these peptide sequences revealed identities with proteins from various Gram-negative bacteria, including iron-regulated OMPs, transporter proteins, as well as TonB-dependent receptors. Taken together, our data suggest that A. pleuropneumoniae synthesizes potential Hm- and Hb-binding proteins that could be implicated in the iron uptake from porcine Hb and Hm.
Figures

Similar articles
-
Evaluation of the hemoglobin-binding activity of Actinobacillus pleuropneumoniae using fluorescein-labeled pig hemoglobin and flow cytometry.FEMS Microbiol Lett. 1999 Apr 1;173(1):17-25. doi: 10.1111/j.1574-6968.1999.tb13479.x. FEMS Microbiol Lett. 1999. PMID: 10220876
-
Fhua and HgbA, outer membrane proteins of Actinobacillus pleuropneumoniae: their role as virulence determinants.Can J Microbiol. 2006 Apr;52(4):391-6. doi: 10.1139/w05-135. Can J Microbiol. 2006. PMID: 16699590
-
Comparative study of iron acquisition by biotype 1 and biotype 2 strains of Actinobacillus pleuropneumoniae.Vet Microbiol. 1995 Apr;44(1):11-23. doi: 10.1016/0378-1135(94)00109-a. Vet Microbiol. 1995. PMID: 7667902
-
Effect of iron regulation on expression and hemin-binding function of outer-sheath proteins from Treponema denticola.Microb Pathog. 1994 May;16(5):321-35. doi: 10.1006/mpat.1994.1033. Microb Pathog. 1994. PMID: 7815916
-
Iron acquisition in the dental pathogen Actinobacillus actinomycetemcomitans: what does it use as a source and how does it get this essential metal?Biometals. 2007 Jun;20(3-4):365-77. doi: 10.1007/s10534-006-9058-3. Epub 2007 Jan 6. Biometals. 2007. PMID: 17206384 Review.
Cited by
-
HbpA from Glaesserella parasuis induces an inflammatory response in 3D4/21 cells by activating the MAPK and NF-κB signalling pathways and protects mice against G. parasuis when used as an immunogen.Vet Res. 2024 Jul 29;55(1):93. doi: 10.1186/s13567-024-01344-4. Vet Res. 2024. PMID: 39075605 Free PMC article.
-
Transcriptional profiling of Actinobacillus pleuropneumoniae under iron-restricted conditions.BMC Genomics. 2007 Mar 13;8:72. doi: 10.1186/1471-2164-8-72. BMC Genomics. 2007. PMID: 17355629 Free PMC article.
-
Surface polysaccharides and iron-uptake systems of Actinobacillus pleuropneumoniae.Can J Vet Res. 2004 Apr;68(2):81-5. Can J Vet Res. 2004. PMID: 15188950 Free PMC article. Review.
-
Catecholamines promote Actinobacillus pleuropneumoniae growth by regulating iron metabolism.PLoS One. 2015 Apr 7;10(4):e0121887. doi: 10.1371/journal.pone.0121887. eCollection 2015. PLoS One. 2015. PMID: 25849041 Free PMC article.
References
-
- Lee BC. Quelling the red menace: haem capture by bacteria. Mol Microbiol 1995;18:383–390. - PubMed
-
- Otto BR, Verweij-van Vught JJ, MacLaren DM. Transferrins and heme-compounds as iron sources for pathogenic bacteria. Crit Rev Microbiol 1992;18:217–233. - PubMed
-
- Cornelissen CN, Sparling PF. Iron piracy: acquisition of transferrin- bound iron by bacterial pathogens. Mol Microbiol 1994;14:843–850. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
