Import of the peroxisomal targeting signal type 2 protein 3-ketoacyl-coenzyme a thiolase into glyoxysomes
- PMID: 14630959
- PMCID: PMC300750
- DOI: 10.1104/pp.103.028217
Import of the peroxisomal targeting signal type 2 protein 3-ketoacyl-coenzyme a thiolase into glyoxysomes
Abstract
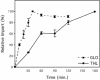
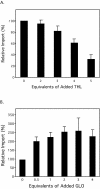
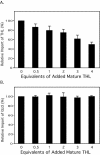
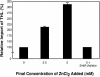
Most peroxisomal matrix proteins possess a carboxy-terminal tripeptide targeting signal, termed peroxisomal targeting signal type 1 (PTS1), and follow a relatively well-characterized pathway of import into the organelle. The peroxisomal targeting signal type 2 (PTS2) pathway of peroxisomal matrix protein import is less well understood. In this study, we investigated the mechanisms of PTS2 protein binding and import using an optimized in vitro assay to reconstitute the transport events. The import of the PTS2 protein thiolase differed from PTS1 protein import in several ways. Thiolase import was slower than typical PTS1 protein import. Competition experiments with both PTS1 and PTS2 proteins revealed that PTS2 protein import was inhibited by addition of excess PTS2 protein, but it was enhanced by the addition of PTS1 proteins. Mature thiolase alone, lacking the PTS2 signal, was not imported into peroxisomes, confirming that the PTS2 signal is necessary for thiolase import. In competition experiments, mature thiolase did not affect the import of a PTS1 protein, but it did decrease the amount of radiolabeled full-length thiolase that was imported. This is consistent with a mechanism by which the mature protein competes with the full-length thiolase during assembly of an import complex at the surface of the membrane. Finally, the addition of zinc to PTS2 protein imports increased the level of thiolase bound and imported into the organelles.
Figures



Similar articles
-
Mutational analyses of a type 2 peroxisomal targeting signal that is capable of directing oligomeric protein import into tobacco BY-2 glyoxysomes.Plant J. 1998 Dec;16(6):709-20. doi: 10.1046/j.1365-313x.1998.00344.x. Plant J. 1998. PMID: 10069077
-
The import receptor for the peroxisomal targeting signal 2 (PTS2) in Saccharomyces cerevisiae is encoded by the PAS7 gene.EMBO J. 1996 Jun 17;15(12):2901-13. EMBO J. 1996. PMID: 8670791 Free PMC article.
-
Pex20p functions as the receptor for non-PTS1/non-PTS2 acyl-CoA oxidase import into peroxisomes of the yeast Yarrowia lipolytica.Traffic. 2019 Jul;20(7):504-515. doi: 10.1111/tra.12652. Epub 2019 May 27. Traffic. 2019. PMID: 31042004
-
The type-2 peroxisomal targeting signal.Biochim Biophys Acta Mol Cell Res. 2020 Feb;1867(2):118609. doi: 10.1016/j.bbamcr.2019.118609. Epub 2019 Nov 18. Biochim Biophys Acta Mol Cell Res. 2020. PMID: 31751594 Review.
-
Targeted fluorescent probes in peroxisome function.Histochem J. 2001 Feb;33(2):65-9. doi: 10.1023/a:1017927728892. Histochem J. 2001. PMID: 11432641 Review.
Cited by
-
Translating the Arabidopsis thaliana Peroxisome Proteome Insights to Solanum lycopersicum: Consensus Versus Diversity.Front Cell Dev Biol. 2022 Jul 13;10:909604. doi: 10.3389/fcell.2022.909604. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35912119 Free PMC article. Review.
-
The Arabidopsis peroxisomal targeting signal type 2 receptor PEX7 is necessary for peroxisome function and dependent on PEX5.Mol Biol Cell. 2005 Feb;16(2):573-83. doi: 10.1091/mbc.e04-05-0422. Epub 2004 Nov 17. Mol Biol Cell. 2005. PMID: 15548601 Free PMC article.
-
PiSCP1 and PiCDPK2 Localize to Peroxisomes and Are Involved in Pollen Tube Growth in Petunia Inflata.Plants (Basel). 2013 Mar 4;2(1):72-86. doi: 10.3390/plants2010072. Plants (Basel). 2013. PMID: 27137367 Free PMC article.
-
Import mechanism of peroxisomal proteins with an N-terminal signal sequence.Nat Cell Biol. 2025 Jun;27(6):948-958. doi: 10.1038/s41556-025-01662-5. Epub 2025 May 9. Nat Cell Biol. 2025. PMID: 40346349 Free PMC article.
-
Covalent Label Transfer between Peroxisomal Importomer Components Reveals Export-driven Import Interactions.J Biol Chem. 2016 Jan 29;291(5):2460-8. doi: 10.1074/jbc.M115.686501. Epub 2015 Nov 13. J Biol Chem. 2016. PMID: 26567336 Free PMC article.
References
-
- Behari R, Baker A (1993) The carboxyl terminus of isocitrate lyase is not essential for import into glyoxysomes in an in vitro system. J Biol Chem 268: 7315-7322 - PubMed
-
- Crookes WJ, Olsen LJ (1998) The effects of chaperones and the influence of protein assembly on peroxisomal protein import. J Biol Chem 273: 17236-17242 - PubMed
-
- Dammai V, Subramani S (2001) The human peroxisomal targeting signal receptor, Pex5p, is translocated into the peroxisomal matrix and recycled to the cytosol. Cell 105: 187-196 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

