Effect of MyBP-C binding to actin on contractility in heart muscle
- PMID: 14638934
- PMCID: PMC2229591
- DOI: 10.1085/jgp.200308941
Effect of MyBP-C binding to actin on contractility in heart muscle
Abstract
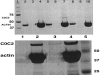
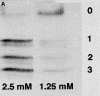
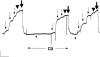
In contrast to skeletal muscle isoforms of myosin binding protein C (MyBP-C), the cardiac isoform has 11 rather than 10 fibronectin or Ig modules (modules are identified as C0 to C10, NH2 to COOH terminus), 3 phosphorylation sites between modules C1 and C2, and 28 additional amino acids rich in proline in C5. Phosphorylation between C1 and C2 increases maximum Ca-activated force (Fmax), alters thick filament structure, and increases the probability of myosin heads on the thick filament binding to actin on the thin filament. Unphosphorylated C1C2 fragment binds to myosin, but phosphorylation inhibits the binding. MyBP-C also binds to actin. Using two types of immunoprecipitation and cosedimentation, we show that fragments of MyBP-C containing C0 bind to actin. In low concentrations C0-containing fragments bind to skinned fibers when the NH2 terminus of endogenous MyBP-C is bound to myosin, but not when MyBP-C is bound to actin. C1C2 fragments bind to skinned fibers when endogenous MyBP-C is bound to actin but not to myosin. Disruption of interactions of endogenous C0 with a high concentration of added C0C2 fragments produces the same effect on contractility as extraction of MyBP-C, namely decrease in Fmax and increase in Ca sensitivity. These results suggest that cardiac contractility can be regulated by shifting the binding of the NH2 terminus of MyBP-C between actin and myosin. This mechanism may have an effect on diastolic filling of the heart.
Figures







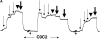

References
-
- Bennett, P., R. Craig, R. Starr, and G. Offer. 1986. The ultrastructural location of C-protein, X-protein and H-protein in rabbit muscle. J. Muscle Res. Cell Motil. 7:550–567. - PubMed
-
- Bonne, G., L. Carrier, J. Bercovici, C. Cruard, P. Richard, B. Hainque, M. Gautel, S. Labiet, M. James, J. Beckman, et al. 1995. Cardiac myosin binding protein C gene splice acceptor site mutation is associated with familial hypertrophic cardiomyopathy. Nat. Genet. 11:438–440. - PubMed
-
- Craig, R., and G. Offer. 1976. The location of C-protein in rabbit skeletal muscle. Proc. R. Soc. Lond. B. Biol. Sci. 192:451–461. - PubMed
-
- Davis, J. 1988. Interaction of C-protein with pH 8.0 synthetic thick filaments prepared from the myosin of vertebrate skeletal muscle. J. Muscle Res. Cell Motil. 9:174–183. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

