Infection and injury of neurons by West Nile encephalitis virus
- PMID: 14645577
- PMCID: PMC296085
- DOI: 10.1128/jvi.77.24.13203-13213.2003
Infection and injury of neurons by West Nile encephalitis virus
Abstract
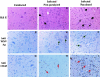
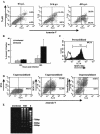
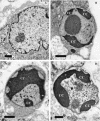
West Nile virus (WNV) infects neurons and leads to encephalitis, paralysis, and death in humans, animals, and birds. We investigated the mechanism by which neuronal injury occurs after WNV infection. Neurons in the anterior horn of the spinal cords of paralyzed mice exhibited a high degree of WNV infection, leukocyte infiltration, and degeneration. Because it was difficult to distinguish whether neuronal injury was caused by viral infection or by the immune system response, a novel tissue culture model for WNV infection was established in neurons derived from embryonic stem (ES) cells. Undifferentiated ES cells were relatively resistant to WNV infection. After differentiation, ES cells expressed neural antigens, acquired a neuronal phenotype, and became permissive for WNV infection. Within 48 h of exposure to an exceedingly low multiplicity of infection (5 x 10(-4)), 50% of ES cell-derived neurons became infected, producing nearly 10(7) PFU of infectious virus per ml, and began to die by an apoptotic mechanism. The establishment of a tractable virus infection model in ES cell-derived neurons facilitates the study of the molecular basis of neurotropism and the mechanisms of viral and immune-mediated neuronal injury after infection by WNV or other neurotropic pathogens.
Figures
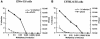


Similar articles
-
The Interferon-Stimulated Gene Ifi27l2a Restricts West Nile Virus Infection and Pathogenesis in a Cell-Type- and Region-Specific Manner.J Virol. 2015 Dec 23;90(5):2600-15. doi: 10.1128/JVI.02463-15. J Virol. 2015. PMID: 26699642 Free PMC article.
-
The Israeli strain IS-98-ST1 of West Nile virus as viral model for West Nile encephalitis in the Old World.Virol J. 2004 Nov 18;1:9. doi: 10.1186/1743-422X-1-9. Virol J. 2004. PMID: 15550172 Free PMC article.
-
Caspase 3-dependent cell death of neurons contributes to the pathogenesis of West Nile virus encephalitis.J Virol. 2007 Mar;81(6):2614-23. doi: 10.1128/JVI.02311-06. Epub 2006 Dec 27. J Virol. 2007. PMID: 17192305 Free PMC article.
-
New insights on the neuropathology of West Nile virus.FEMS Microbiol Lett. 2004 Apr 1;233(1):1-6. doi: 10.1016/j.femsle.2004.01.035. FEMS Microbiol Lett. 2004. PMID: 15098543 Review.
-
West Nile virus and kidney disease.Expert Rev Anti Infect Ther. 2013 May;11(5):479-87. doi: 10.1586/eri.13.34. Expert Rev Anti Infect Ther. 2013. PMID: 23627854 Review.
Cited by
-
Altered protein networks and cellular pathways in severe west nile disease in mice.PLoS One. 2013 Jul 10;8(7):e68318. doi: 10.1371/journal.pone.0068318. Print 2013. PLoS One. 2013. PMID: 23874584 Free PMC article.
-
Zika virus infection causes temporary paralysis in adult mice with motor neuron synaptic retraction and evidence for proximal peripheral neuropathy.Sci Rep. 2019 Dec 20;9(1):19531. doi: 10.1038/s41598-019-55717-3. Sci Rep. 2019. PMID: 31862897 Free PMC article.
-
In vitro and in silico Models to Study Mosquito-Borne Flavivirus Neuropathogenesis, Prevention, and Treatment.Front Cell Infect Microbiol. 2019 Jul 9;9:223. doi: 10.3389/fcimb.2019.00223. eCollection 2019. Front Cell Infect Microbiol. 2019. PMID: 31338335 Free PMC article. Review.
-
Expression and evolutionary analysis of West Nile virus (Merion strain).J Neurovirol. 2005 Dec;11(6):544-56. doi: 10.1080/13550280500385229. J Neurovirol. 2005. PMID: 16338748
-
CD8+ T cells require perforin to clear West Nile virus from infected neurons.J Virol. 2006 Jan;80(1):119-29. doi: 10.1128/JVI.80.1.119-129.2006. J Virol. 2006. PMID: 16352536 Free PMC article.
References
-
- Bain, G., D. Kitchens, M. Yao, J. E. Huettner, and D. I. Gottlieb. 1995. Embryonic stem cells express neuronal properties in vitro. Dev. Biol. 168:342-357. - PubMed
-
- Bauer, J., C. G. Bien, and H. Lassmann. 2002. Rasmussen's encephalitis: a role for autoimmune cytotoxic T lymphocytes. Curr. Opin. Neurol. 15:197-200. - PubMed
-
- Bauer, J., T. Sminia, F. G. Wouterlood, and C. D. Dijkstra. 1994. Phagocytic activity of macrophages and microglial cells during the course of acute and chronic relapsing experimental autoimmune encephalomyelitis. J. Neurosci. Res. 38:365-375. - PubMed
-
- Bernard, K. A., and L. D. Kramer. 2001. West Nile virus activity in the United States, 2001. Viral Immunol. 14:319-338. - PubMed
-
- Bien, C. G., J. Bauer, T. L. Deckwerth, H. Wiendl, M. Deckert, O. D. Wiestler, J. Schramm, C. E. Elger, and H. Lassmann. 2002. Destruction of neurons by cytotoxic T cells: a new pathogenic mechanism in Rasmussen's encephalitis. Ann. Neurol. 51:311-318. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

