Human immunodeficiency virus type 1 DNA nuclear import and integration are mitosis independent in cycling cells
- PMID: 14645598
- PMCID: PMC296078
- DOI: 10.1128/jvi.77.24.13412-13417.2003
Human immunodeficiency virus type 1 DNA nuclear import and integration are mitosis independent in cycling cells
Abstract
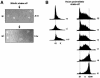
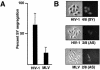
An essential step in human immunodeficiency virus type 1 (HIV-1) replication is the movement of the viral preintegration complex from the cytoplasm into the nucleus. The pathway(s) and timing for HIV-1 DNA nuclear entry in cycling cells have not been established. Here, we show that if cycling cells are infected before S phase, viral DNA can be integrated prior to passage of the host DNA replication fork through the integration site, as indicated by stable inheritance in both daughter cells. We conclude that efficient nuclear entry can occur independently of mitotic nuclear disassembly in cycling cells.
Figures
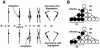

Similar articles
-
Nuclear localization of human immunodeficiency virus type 1 preintegration complexes (PICs): V165A and R166A are pleiotropic integrase mutants primarily defective for integration, not PIC nuclear import.J Virol. 2002 Nov;76(21):10598-607. doi: 10.1128/jvi.76.21.10598-10607.2002. J Virol. 2002. PMID: 12368302 Free PMC article.
-
Active nuclear import of human immunodeficiency virus type 1 preintegration complexes.Proc Natl Acad Sci U S A. 1992 Jul 15;89(14):6580-4. doi: 10.1073/pnas.89.14.6580. Proc Natl Acad Sci U S A. 1992. PMID: 1631159 Free PMC article.
-
Nuclear import of the preintegration complex is blocked upon infection by human immunodeficiency virus type 1 in mouse cells.J Virol. 2007 Jan;81(2):677-88. doi: 10.1128/JVI.00870-06. Epub 2006 Nov 1. J Virol. 2007. PMID: 17079325 Free PMC article.
-
The road to chromatin - nuclear entry of retroviruses.Nat Rev Microbiol. 2007 Mar;5(3):187-96. doi: 10.1038/nrmicro1579. Nat Rev Microbiol. 2007. PMID: 17304248 Review.
-
Viral and cellular requirements for the nuclear entry of retroviral preintegration nucleoprotein complexes.Viruses. 2013 Oct 7;5(10):2483-511. doi: 10.3390/v5102483. Viruses. 2013. PMID: 24103892 Free PMC article. Review.
Cited by
-
Analysis of the viral elements required in the nuclear import of HIV-1 DNA.J Virol. 2010 Jan;84(2):729-39. doi: 10.1128/JVI.01952-09. Epub 2009 Nov 4. J Virol. 2010. PMID: 19889772 Free PMC article.
-
The cell cycle independence of HIV infections is not determined by known karyophilic viral elements.PLoS Pathog. 2005 Nov;1(3):e18. doi: 10.1371/journal.ppat.0010018. Epub 2005 Nov 11. PLoS Pathog. 2005. PMID: 16292356 Free PMC article.
-
HIV integration targeting: a pathway involving Transportin-3 and the nuclear pore protein RanBP2.PLoS Pathog. 2011 Mar;7(3):e1001313. doi: 10.1371/journal.ppat.1001313. Epub 2011 Mar 10. PLoS Pathog. 2011. PMID: 21423673 Free PMC article.
-
HIV-1 viral protein R (Vpr) and its interactions with host cell.Curr HIV Res. 2009 Mar;7(2):178-83. doi: 10.2174/157016209787581436. Curr HIV Res. 2009. PMID: 19275587 Free PMC article. Review.
-
ATP generation in a host cell in early-phase infection is increased by upregulation of cytochrome c oxidase activity via the p2 peptide from human immunodeficiency virus type 1 Gag.Retrovirology. 2015 Nov 17;12:97. doi: 10.1186/s12977-015-0224-y. Retrovirology. 2015. PMID: 26577226 Free PMC article.
References
-
- Bouyac-Bertoia, M., J. D. Dvorin, R. A. Fouchier, Y. Jenkins, B. E. Meyer, L. I. Wu, M. Emerman, and M. H. Malim. 2001. HIV-1 infection requires a functional integrase NLS. Mol. Cell 7:1025-1035. - PubMed
-
- Butler, S. L., M. S. Hansen, and F. D. Bushman. 2001. A quantitative assay for HIV DNA integration in vivo. Nat. Med. 7:631-634. - PubMed
-
- Chen, W., X. Wu, D. N. Levasseur, H. Liu, L. Lai, J. C. Kappes, and T. M. Townes. 2000. Lentiviral vector transduction of hematopoietic stem cells that mediate long-term reconstitution of lethally irradiated mice. Stem Cells 18:352-359. - PubMed
-
- Coffin, J. M., S. H. Hughes, and H. Varmus. 1997. Retroviruses. Cold Spring Harbor Laboratory Press, Plainview, N.Y. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

