Remote thermometry to avoid complications in radiofrequency ablation
- PMID: 14654495
- PMCID: PMC2408961
- DOI: 10.1097/01.rvi.0000096769.74047.5
Remote thermometry to avoid complications in radiofrequency ablation
Abstract
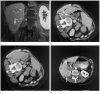
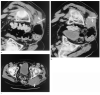
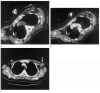
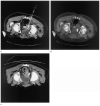
Percutaneous image-guided radiofrequency ablation (RFA) of tumors has the potential risk for thermal damage to nearby normal collateral tissues. Thus, the goal of creating a sufficient area of tumor necrosis must be weighed against the risk for injury to collateral tissues. In this study, remote thermistors were used to monitor temperatures near collateral structures during tumor RFA. Four unique cases are described. When temperature-sensitive structures are near the target lesion, remote thermometry could further increase the safety of this evolving minimally invasive procedure.
Figures


References
-
- Nahum Goldberg S, Dupuy DE. Image-guided radiofrequency tumor ablation: challenges and opportunities–part I. J Vasc Interv Radiol. 2001;12:1021–1032. - PubMed
-
- Dewey WC. Arrhenius relationships from the molecule and cell to the clinic. Int J Hyperthermia. 1994;10:457–483. - PubMed
-
- Dupuy DE, Hong R, Oliver B, Goldberg SN. Radiofrequency ablation of spinal tumors: temperature distribution in the spinal canal. AJR Am J Roentgenol. 2000;175:1263–1266. - PubMed
-
- Yamane T, Tateishi A, Cho S, et al. The effects of hyperthermia on the spinal cord. Spine. 1992;17:1386–1391. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
