Individual properties of the two functional agonist sites in GABA(A) receptors
- PMID: 14657175
- PMCID: PMC6741049
- DOI: 10.1523/JNEUROSCI.23-35-11158.2003
Individual properties of the two functional agonist sites in GABA(A) receptors
Erratum in
- J Neurosci. 2003 Dec 17;23(37):11778
Abstract
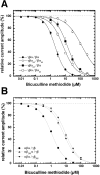
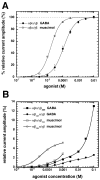
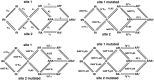
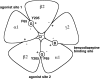
The members of the pentameric ligand-gated receptor channel family are involved in information transfer in synapses and the neuromuscular junction. They often contain several copies of the same subunit isoform. Here, we present a method to functionally dissect the role of individual subunits that occur in multiple copies in these receptors. Opening of the inherent chloride channel in the GABA(A) receptor is achieved through the binding of two agonist molecules; however, it has been difficult to obtain information on the contribution of the two individual binding sites. The sites are both located at beta+/alpha- subunit interfaces, suggesting similar properties. One pair of subunits is flanked by gamma and beta (site 1) and the other by alpha and gamma (site 2), the different environment possibly affecting the binding sites. Here, we used concatenated subunits and two point mutations of amino acid residues, each in alpha and beta subunits, both located in the agonist binding pocket, to investigate the properties of these two sites. The sites were individually mutated, and consequences of these mutations on GABA and muscimol-induced channel opening and its competitive inhibition by bicuculline were studied. A model predicts that opening also occurs for receptors occupied with a single agonist molecule but is promoted approximately 60-fold in those occupied by two agonists and that site 2 has an approximately threefold higher affinity for GABA than site 1, whereas muscimol and bicuculline show some preference for site 1.
Figures



Similar articles
-
On high- and low-affinity agonist sites in GABAA receptors.J Neurochem. 2003 Oct;87(2):325-32. doi: 10.1046/j.1471-4159.2003.01982.x. J Neurochem. 2003. PMID: 14511110
-
Bicuculline and gabazine are allosteric inhibitors of channel opening of the GABAA receptor.J Neurosci. 1997 Jan 15;17(2):625-34. doi: 10.1523/JNEUROSCI.17-02-00625.1997. J Neurosci. 1997. PMID: 8987785 Free PMC article.
-
Structure and dynamics of the GABA binding pocket: A narrowing cleft that constricts during activation.J Neurosci. 2001 Jan 1;21(1):67-74. doi: 10.1523/JNEUROSCI.21-01-00067.2001. J Neurosci. 2001. PMID: 11150321 Free PMC article.
-
A closer look at the high affinity benzodiazepine binding site on GABAA receptors.Curr Top Med Chem. 2011;11(2):241-6. doi: 10.2174/156802611794863562. Curr Top Med Chem. 2011. PMID: 21189125 Review.
-
Mapping of the benzodiazepine recognition site on GABA(A) receptors.Curr Top Med Chem. 2002 Aug;2(8):833-9. doi: 10.2174/1568026023393444. Curr Top Med Chem. 2002. PMID: 12171574 Review.
Cited by
-
Do N-arachidonyl-glycine (NA-glycine) and 2-arachidonoyl glycerol (2-AG) share mode of action and the binding site on the β2 subunit of GABAA receptors?PeerJ. 2013 Sep 10;1:e149. doi: 10.7717/peerj.149. eCollection 2013. PeerJ. 2013. PMID: 24058880 Free PMC article.
-
Probes for the heterogeneity of muscimol binding sites in rat brain.Front Pharmacol. 2024 Mar 14;15:1368527. doi: 10.3389/fphar.2024.1368527. eCollection 2024. Front Pharmacol. 2024. PMID: 38549678 Free PMC article.
-
Novel Molecule Exhibiting Selective Affinity for GABAA Receptor Subtypes.Sci Rep. 2017 Jul 24;7(1):6230. doi: 10.1038/s41598-017-05966-x. Sci Rep. 2017. PMID: 28740086 Free PMC article.
-
The point mutation gamma 2F77I changes the potency and efficacy of benzodiazepine site ligands in different GABAA receptor subtypes.Eur J Pharmacol. 2010 Jun 25;636(1-3):18-27. doi: 10.1016/j.ejphar.2010.03.015. Epub 2010 Mar 19. Eur J Pharmacol. 2010. PMID: 20303942 Free PMC article.
-
Positioning of the alpha-subunit isoforms confers a functional signature to gamma-aminobutyric acid type A receptors.Proc Natl Acad Sci U S A. 2004 May 18;101(20):7769-74. doi: 10.1073/pnas.0400220101. Epub 2004 May 10. Proc Natl Acad Sci U S A. 2004. PMID: 15136735 Free PMC article.
References
-
- Amin J, Weiss DS ( 1993) GABAA receptor needs two homologous domains of the β-subunit for activation by GABA but not by pentobarbital. Nature 366: 565-569. - PubMed
-
- Amin J, Brooks-Kayal A, Weiss DS ( 1997) Two tyrosine residues on the α subunit are crucial for benzodiazepine binding and allosteric modulation of gamma-aminobutyric acidA receptors. Mol Pharmacol 51: 833-841. - PubMed
-
- Backus KH, Arigoni M, Drescher U, Scheurer L, Malherbe P, Möhler H, Benson JA ( 1993) Stoichiometry of a recombinant GABAA receptor deduced from mutation-induced rectification. NeuroReport 5: 285-288. - PubMed
-
- Barnard EA, Skolnick P, Olsen RW, Möhler H, Sieghart W, Biggio G, Braestrup C, Bateson AN, Langer SZ ( 1998) International union of pharmacology. XV. Subtypes of gamma-aminobutyric acidA receptors: classification on the basis of subunit structure and receptor function. Pharmacol Rev 50: 291-313. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
