Subsets of memory cytotoxic T lymphocytes elicited by vaccination influence the efficiency of secondary expansion in vivo
- PMID: 14671102
- PMCID: PMC303428
- DOI: 10.1128/jvi.78.1.206-215.2004
Subsets of memory cytotoxic T lymphocytes elicited by vaccination influence the efficiency of secondary expansion in vivo
Abstract
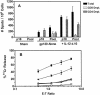
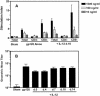
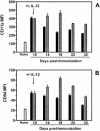
Vaccine-elicited cytotoxic T lymphocytes (CTL) should be long-lived memory cells that can rapidly expand in number following re-exposure to antigen. The present studies were initiated to analyze the ability of plasmid interleukin-12 (IL-12) to augment CTL responses in mice when delivered during the peak phase of an immune response elicited by a plasmid human immunodeficiency virus type 1 gp120 DNA vaccine. Delivery of plasmid IL-12 on day 10 postimmunization resulted in a robust expansion of gp120-specific CD8+ T cells, as measured by tetramer, gamma interferon ELISPOT, and functional-killing assays. Interestingly, this delayed administration of plasmid IL-12 had no significant effect on antigen-specific CD4(+)-T-cell and antibody responses. Phenotypic analyses suggested that administration of plasmid IL-12 near the time of the peak CTL response activated and expanded antigen-specific effector cells, preventing their loss through apoptosis. However, this IL-12-augmented population of gp120-specific CD8+ T cells did not efficiently expand following gp120 boost immunization, suggesting that these effector cells would be of little utility in expanding to contain a viral infection. Analyses of the phenotypic profile and anatomic distribution of the plasmid IL-12-augmented CTL population indicated that these lymphocytes were primarily effector memory rather than central memory T cells. These observations suggest that CTL-based vaccines should elicit central memory rather than effector memory T cells and illustrate the importance of monitoring the phenotype and functionality of vaccine-induced, antigen-specific CTL.
Figures


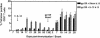
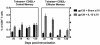
References
-
- Barouch, D. H., S. Santra, M. J. Kuroda, J. E. Schmitz, R. Plishka, A. Buckler-White, A. E. Gaitan, R. Zin, J.-H. Nam, L. S. Wyatt, M. A. Lifton, C. E. Nickerson, B. Moss, D. C. Montefiori, V. M. Hirsch, and N. L. Letvin. 2001. Reduction of simian-human immunodeficiency virus 89.6P viremia in rhesus monkeys by recombinant modified vaccinia virus Ankara vaccination. J. Virol. 75:5151-5158. - PMC - PubMed
-
- Barouch, D. H., S. Santra, J. E. Schmitz, M. J. Kuroda, T. M. Fu, W. Wagner, M. Bilska, A. Craiu, X. X. Zheng, G. R. Krivulka, K. Beaudry, M. A. Lifton, C. E. Nickerson, W. L. Trigona, K. Punt, D. C. Freed, L. Guan, S. Dubey, D. Casimiro, A. Simon, M. E. Davies, M. Chastain, T. B. Strom, R. S. Gelman, D. C. Montefiori, M. G. Lewis, E. A. Emini, J. W. Shiver, and N. L. Letvin. 2000. Control of viremia and prevention of clinical AIDS in rhesus monkeys by cytokine-augmented DNA vaccination. Science 290:486-492. - PubMed
-
- Barouch, D. H., S. Santra, T. D. Steenbeke, X. X. Zheng, H. C. Perry, M. E. Davies, D. C. Freed, A. Craiu, T. B. Strom, J. W. Shiver, and N. L. Letvin. 1998. Augmentation and suppression of immune responses to an HIV-1 DNA vaccine by plasmid cytokine/Ig administration. J. Immunol. 161:1875-1882. - PubMed
-
- Calarota, S., G. Bratt, S. Nordlund, J. Hinkula, A. C. Leandersson, E. Sandstrom, and B. Wahren. 1998. Cellular cytotoxic response induced by DNA vaccination in HIV-1-infected patients. Lancet 351:1320-1325. - PubMed
-
- Chen, H. W., C. H. Pan, H. W. Huan, M. Y. Liau, J. R. Chiang, and M. H. Tao. 2001. Suppression of immune response and protective immunity to a Japanese encephalitis virus DNA vaccine by coadministration of an IL-12-expressing plasmid. J. Immunol. 166:7419-7426. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

