A JC virus-induced signal is required for infection of glial cells by a clathrin- and eps15-dependent pathway
- PMID: 14671106
- PMCID: PMC303400
- DOI: 10.1128/jvi.78.1.250-256.2004
A JC virus-induced signal is required for infection of glial cells by a clathrin- and eps15-dependent pathway
Erratum in
-
Correction for Querbes et al., "A JC Virus-Induced Signal Is Required for Infection of Glial Cells by a Clathrin- and eps15-Dependent Pathway".J Virol. 2024 Apr 16;98(4):e0028224. doi: 10.1128/jvi.00282-24. Epub 2024 Mar 26. J Virol. 2024. PMID: 38529953 Free PMC article. No abstract available.
Abstract
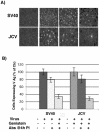
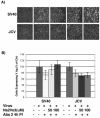
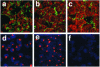
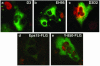
Infectious entry of JC virus (JCV) into human glial cells occurs by receptor-mediated clathrin-dependent endocytosis. In this report we demonstrate that the tyrosine kinase inhibitor genistein blocks virus entry and inhibits infection. Transient expression of dominant-negative eps15 mutants, including a phosphorylation-defective mutant, inhibited both virus entry and infection. We also show that the JCV-induced signal activates the mitogen-activated protein kinases ERK1 and ERK2. These data demonstrate that JC virus binding to human glial cells induces an intracellular signal that is critical for entry and infection by a ligand-inducible clathrin-dependent mechanism.
Figures


Similar articles
-
JC virus enters human glial cells by clathrin-dependent receptor-mediated endocytosis.J Virol. 2000 Mar;74(5):2288-92. doi: 10.1128/jvi.74.5.2288-2292.2000. J Virol. 2000. PMID: 10666259 Free PMC article.
-
Ligand-induced clathrin-mediated endocytosis of the keratinocyte growth factor receptor occurs independently of either phosphorylation or recruitment of eps15.FEBS Lett. 2003 Oct 23;553(3):262-70. doi: 10.1016/s0014-5793(03)01020-2. FEBS Lett. 2003. PMID: 14572635
-
Mouse hepatitis virus type 2 enters cells through a clathrin-mediated endocytic pathway independent of Eps15.J Virol. 2008 Aug;82(16):8112-23. doi: 10.1128/JVI.00837-08. Epub 2008 Jun 11. J Virol. 2008. PMID: 18550663 Free PMC article.
-
Epidermal growth factor pathway substrate 15, Eps15.Int J Biochem Cell Biol. 1999 Aug;31(8):805-9. doi: 10.1016/s1357-2725(99)00042-4. Int J Biochem Cell Biol. 1999. PMID: 10481267 Review.
-
Molecular mechanisms underlying endocytosis and sorting of ErbB receptor tyrosine kinases.FEBS Lett. 2001 Feb 16;490(3):142-52. doi: 10.1016/s0014-5793(01)02117-2. FEBS Lett. 2001. PMID: 11223029 Review.
Cited by
-
Japanese encephalitis virus infects porcine kidney epithelial PK15 cells via clathrin- and cholesterol-dependent endocytosis.Virol J. 2013 Aug 12;10:258. doi: 10.1186/1743-422X-10-258. Virol J. 2013. PMID: 23937769 Free PMC article.
-
CRISPR antiviral inhibits neurotrophic JC polyomavirus in 2D and 3D culture models through dual-gRNA excision by SaCas9.Mol Ther Nucleic Acids. 2025 May 14;36(2):102556. doi: 10.1016/j.omtn.2025.102556. eCollection 2025 Jun 10. Mol Ther Nucleic Acids. 2025. PMID: 40510594 Free PMC article.
-
Adipocyte Plasma Membrane Protein (APMAP) promotes JC Virus (JCPyV) infection in human glial cells.Virology. 2020 Sep;548:17-24. doi: 10.1016/j.virol.2020.06.002. Epub 2020 Jun 7. Virology. 2020. PMID: 32838939 Free PMC article.
-
Rearrangement in the Hypervariable Region of JC Polyomavirus Genomes Isolated from Patient Samples and Impact on Transcription Factor-Binding Sites and Disease Outcomes.Int J Mol Sci. 2022 May 20;23(10):5699. doi: 10.3390/ijms23105699. Int J Mol Sci. 2022. PMID: 35628509 Free PMC article.
-
The oncogenic roles of JC polyomavirus in cancer.Front Oncol. 2022 Sep 23;12:976577. doi: 10.3389/fonc.2022.976577. eCollection 2022. Front Oncol. 2022. PMID: 36212474 Free PMC article. Review.
References
-
- Antinori, A., A. Ammassari, M. L. Giancola, A. Cingolani, S. Grisetti, R. Murri, L. Alba, B. Ciancio, F. Soldani, D. Larussa, G. Ippolito, and A. De Luca. 2001. Epidemiology and prognosis of AIDS-associated progressive multifocal leukoencephalopathy in the HAART era. J. Neurovirol. 7:323-328. - PubMed
-
- Benmerah, A., M. Bayrou, N. Cerf-Bensussan, and A. Dautry-Varsat. 1999. Inhibition of clathrin-coated pit assembly by an Eps15 mutant. J. Cell Sci. 112:1303-1311. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

