Immune failure in the absence of profound CD4+ T-lymphocyte depletion in simian immunodeficiency virus-infected rapid progressor macaques
- PMID: 14671109
- PMCID: PMC303408
- DOI: 10.1128/jvi.78.1.275-284.2004
Immune failure in the absence of profound CD4+ T-lymphocyte depletion in simian immunodeficiency virus-infected rapid progressor macaques
Abstract
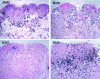
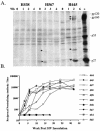
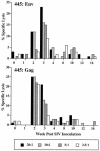
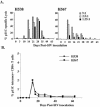
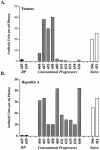
A fraction of simian immunodeficiency virus (SIV)-infected macaques develop rapidly progressive disease in the apparent absence of detectable SIV-specific antibody responses. To characterize the immunopathogenesis of this syndrome, we studied viral load, CD4+ T-lymphocyte numbers as well as cellular and humoral immune responses to SIV and other exogenous antigens in four SIVsm-infected rhesus macaques that progressed to AIDS 9 to 16 weeks postinoculation. Each of these animals exhibited high levels of viremia but showed relatively preserved CD4 T lymphocytes in blood and lymphoid tissues at the time of death. Transient SIV-specific antibody responses and cytotoxic T-lymphocyte responses were observed at 2 to 4 weeks postinoculation. Two of the macaques that were immunized sequentially with tetanus toxoid and hepatitis A virus failed to develop antibody to either antigen. These studies show that the SIV-infected rapid progressor macaques initially mounted an appropriate but transient cellular and humoral immune response. The subsequent immune defect in these animals appeared to be global, affecting both cellular and humoral immunity to SIV as well as immune responses against unrelated antigens. The lack of CD4 depletion and loss of humoral and cellular immune responses suggest that their immune defect may be due to an early loss in T helper function.
Figures
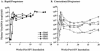
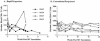

Similar articles
-
Loss of naïve cells accompanies memory CD4+ T-cell depletion during long-term progression to AIDS in Simian immunodeficiency virus-infected macaques.J Virol. 2007 Jan;81(2):893-902. doi: 10.1128/JVI.01635-06. Epub 2006 Nov 8. J Virol. 2007. PMID: 17093193 Free PMC article.
-
Occult systemic infection and persistent simian immunodeficiency virus (SIV)-specific CD4(+)-T-cell proliferative responses in rhesus macaques that were transiently viremic after intravaginal inoculation of SIV.J Virol. 1998 Dec;72(12):10029-35. doi: 10.1128/JVI.72.12.10029-10035.1998. J Virol. 1998. PMID: 9811741 Free PMC article.
-
Differential Impact of In Vivo CD8+ T Lymphocyte Depletion in Controller versus Progressor Simian Immunodeficiency Virus-Infected Macaques.J Virol. 2015 Sep;89(17):8677-86. doi: 10.1128/JVI.00869-15. Epub 2015 Jun 10. J Virol. 2015. PMID: 26063417 Free PMC article.
-
Cellular immune response to SIVmac and HIV-2 in macaques: model for the human HIV-1 infection.J Acquir Immune Defic Syndr (1988). 1993 Sep;6(9):969-76. J Acquir Immune Defic Syndr (1988). 1993. PMID: 8340900 Review.
-
CD4+ T-cell depletion in HIV infection: killed by friendly fire?Curr HIV/AIDS Rep. 2005 Feb;2(1):16-23. doi: 10.1007/s11904-996-0004-3. Curr HIV/AIDS Rep. 2005. PMID: 16091244 Review.
Cited by
-
Simian immunodeficiency virus infects follicular helper CD4 T cells in lymphoid tissues during pathogenic infection of pigtail macaques.J Virol. 2013 Apr;87(7):3760-73. doi: 10.1128/JVI.02497-12. Epub 2013 Jan 16. J Virol. 2013. PMID: 23325697 Free PMC article.
-
Highly pathogenic SHIVs and SIVs target different CD4+ T cell subsets in rhesus monkeys, explaining their divergent clinical courses.Proc Natl Acad Sci U S A. 2004 Aug 17;101(33):12324-9. doi: 10.1073/pnas.0404620101. Epub 2004 Aug 5. Proc Natl Acad Sci U S A. 2004. PMID: 15297611 Free PMC article.
-
Coreceptor use in nonhuman primate models of HIV infection.J Transl Med. 2011 Jan 27;9 Suppl 1(Suppl 1):S7. doi: 10.1186/1479-5876-9-S1-S7. J Transl Med. 2011. PMID: 21284906 Free PMC article. Review.
-
CD4+ T cells support production of simian immunodeficiency virus Env antibodies that enforce CD4-dependent entry and shape tropism in vivo.J Virol. 2013 Sep;87(17):9719-32. doi: 10.1128/JVI.01254-13. Epub 2013 Jul 3. J Virol. 2013. PMID: 23824793 Free PMC article.
-
Persistent transcription of a nonintegrating mutant of simian immunodeficiency virus in rhesus macrophages.Virology. 2008 Mar 15;372(2):291-9. doi: 10.1016/j.virol.2007.11.002. Epub 2007 Dec 3. Virology. 2008. PMID: 18054062 Free PMC article.
References
-
- Barouch, D. H., J. Kunstman, J. Glowczwskie, K. J. Kunstman, M. A. Egan, F. W. Peyer S. Santra, M. J. Kuroda, J. E. Schmitz, K. Beaudry, G. R. Krivulka, M. A. Lifton, D. A. Gorgone, S. M. Wolinsky, and N. L. Letvin. 2003. Viral escape from dominant simian immunodeficiency virus epitope-specific cytotoxic T lymphocytes in DNA-vaccinated rhesus monkeys. J. Virol. 77:7367-7375. - PMC - PubMed
-
- Baskin, G. B., M. Murphey-Corb, E. A. Watson, and L. N. Martin. 1988. Necropsy findings in rhesus monkeys experimentally infected with cultured simian immunodeficiency virus (SIV/Delta). Vet. Pathol. 25:456-467. - PubMed
-
- Cao, Y., L. Qin, L. Zhang, J. Safrit, and D. Ho. 1995. Virologic and immunologic characterization of long-term survivors of human immunodeficiency virus type 1 infection. N. Engl. J. Med. 332:201-208. - PubMed
-
- Carrington, M., G. W. Nelson, M. P. Martin, T. Kissner, D. Vlahov, J. J. Goedert, R. Kaslow, S. Buchbinder, K. Hoots, and S. J. O'Brien. 1999. HLA and HIV-1: heterozygote advantage and B*35-Cw*04 disadvantage. Science 283:1748-1752. - PubMed
-
- Deacon, N. J., A. Tsykin, A. Solomon, K. Smith, M. Ludford-Menting, D. J. Hooker, D. A. McPhee, A. L. Greenway, A. Ellett, C. Chatfield, et al. 1995. Genomic structure of an attenuated quasi species of HIV-1 from a blood transfusion donor and recipients. Science 270:988-991. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

