The Rep protein of adeno-associated virus type 2 interacts with single-stranded DNA-binding proteins that enhance viral replication
- PMID: 14671124
- PMCID: PMC303412
- DOI: 10.1128/jvi.78.1.441-453.2004
The Rep protein of adeno-associated virus type 2 interacts with single-stranded DNA-binding proteins that enhance viral replication
Abstract
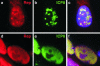
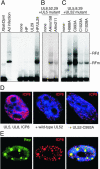
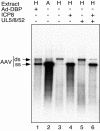
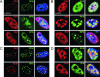
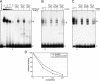
Adeno-associated virus (AAV) type 2 is a human parvovirus whose replication is dependent upon cellular proteins as well as functions supplied by helper viruses. The minimal herpes simplex virus type 1 (HSV-1) proteins that support AAV replication in cell culture are the helicase-primase complex of UL5, UL8, and UL52, together with the UL29 gene product ICP8. We show that AAV and HSV-1 replication proteins colocalize at discrete intranuclear sites. Transfections with mutant genes demonstrate that enzymatic functions of the helicase-primase are not essential. The ICP8 protein alone enhances AAV replication in an in vitro assay. We also show localization of the cellular replication protein A (RPA) at AAV centers under a variety of conditions that support replication. In vitro assays demonstrate that the AAV Rep68 and Rep78 proteins interact with the single-stranded DNA-binding proteins (ssDBPs) of Ad (Ad-DBP), HSV-1 (ICP8), and the cell (RPA) and that these proteins enhance binding and nicking of Rep proteins at the origin. These results highlight the importance of intranuclear localization and suggest that Rep interaction with multiple ssDBPs allows AAV to replicate under a diverse set of conditions.
Figures



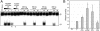
References
-
- Arana, M. E., B. Haq, N. Tanguy Le Gac, and P. E. Boehmer. 2001. Modulation of the herpes simplex virus type-1 UL9 DNA helicase by its cognate single-strand DNA-binding protein, ICP8. J. Biol. Chem. 276:6840-6845. - PubMed
-
- Biswas, N., and S. K. Weller. 1999. A mutation in the C-terminal putative Zn2+ finger motif of UL52 severely affects the biochemical activities of the HSV-1 helicase-primase subcomplex. J. Biol. Chem. 274:8068-8076. - PubMed
-
- Biswas, N., and S. K. Weller. 2001. The UL5 and UL52 subunits of the herpes simplex virus type 1 helicase-primase subcomplex exhibit a complex interdependence for DNA binding. J. Biol. Chem. 276:17610-17619. - PubMed
-
- Boehmer, P. E. 1998. The herpes simplex virus type-1 single-strand DNA-binding protein, ICP8, increases the processivity of the UL9 protein DNA helicase. J. Biol. Chem. 273:2676-2683. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

