A point mutation in the binding subunit of a retroviral envelope protein arrests virus entry at hemifusion
- PMID: 14671127
- PMCID: PMC303374
- DOI: 10.1128/jvi.78.1.473-481.2004
A point mutation in the binding subunit of a retroviral envelope protein arrests virus entry at hemifusion
Abstract
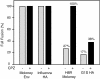
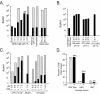
The transmembrane subunits of viral envelope proteins are thought to perform all of the functions required for membrane fusion during entry of enveloped viruses. However, changes in a conserved SPHQ motif near the N terminus of the receptor binding subunit of a murine leukemia virus (MLV) envelope protein block infection and induction of cell-cell fusion but not receptor binding. Here we report evidence that a histidine-to-arginine change at position 8 (H8R) in the SPHQ motif of Moloney MLV blocks infection by arresting virus-cell fusion at the hemifusion state. In cell-cell fusion assays, H8R envelope protein induced mixing of membrane outer leaflet lipids but did not lead to content mixing, a finding indicative of fusion pore formation. Kinetic studies of virus-cell fusion showed that lipid mixing of H8R virus membranes begins much later than for wild-type virus. The length of the delay in lipid mixing decreased upon addition of two second-site changes that increase H8R virus infection to 100-fold less than the wild-type virus. Finally, chlorpromazine, dibucaine, and trifluoperazine, agents that induce pores in an arrested hemifusion state, rescued infection by H8R virus to within 2.5-fold of the level of wild-type virus infection and cell-cell fusion to half that mediated by wild-type envelope protein. We interpret these results to indicate that fusion progressed to the hemifusion intermediate but fusion pore formation was inhibited. These results establish that membrane fusion of Moloney MLV occurs via a hemifusion intermediate. We also interpret these findings as evidence that histidine 8 is a key switch-point residue between the receptor-induced conformation changes that expose fusion peptide and those that lead to six-helix bundle formation.
Figures



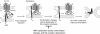
References
-
- Ausubel, F. M., R. Brent, R. E. Kingston, D. D. Moore, J. G. Seidman, J. A. Smith, and K. Struhl (ed.). 1995. Current protocols in molecular biology, vol. 2. John Wiley & Sons, Inc., New York, N.Y.
-
- Bagai, S., and R. A. Lamb. 1997. A glycine to alanine substitution in the paramyxovirus SV5 fusion peptide increases the initial rate of fusion. Virology 238:283-290. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

