Imaging reveals synaptic targets of a swim-terminating neuron in the leech CNS
- PMID: 14673004
- PMCID: PMC6740517
- DOI: 10.1523/JNEUROSCI.23-36-11402.2003
Imaging reveals synaptic targets of a swim-terminating neuron in the leech CNS
Abstract
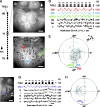
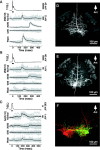
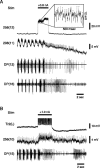
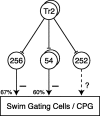
In the leech, the command-like neuron called cell Tr2 is known to stop swimming, but the connections from cell Tr2 to the swim central pattern generator have not been identified. We used fluorescence resonance energy transfer voltage-sensitive dyes to identify three neurons that are synaptic targets of cell Tr2. We then used electrophysiological techniques to show that these connections are monosynaptic, chemical, and excitatory. Two of the novel targets, cell 256 and cell 54, terminate swimming when stimulated. These neurons are likely to mediate swim cessation caused by cell Tr2 activity, and thus play the role of intermediate control cells in the leech CNS.
Figures

Similar articles
-
Termination of leech swimming activity by a previously identified swim trigger neuron.J Comp Physiol A. 1995 Nov;177(5):627-36. doi: 10.1007/BF00207191. J Comp Physiol A. 1995. PMID: 7473307
-
Initiation of swimming activity by trigger neurons in the leech subesophageal ganglion. I. Output connections of Tr1 and Tr2.J Comp Physiol A. 1986 Oct;159(4):489-502. doi: 10.1007/BF00604169. J Comp Physiol A. 1986. PMID: 3783502
-
Identified neurons and leech swimming behavior.Prog Neurobiol. 2001 Mar;63(4):371-81. doi: 10.1016/s0301-0082(00)00048-4. Prog Neurobiol. 2001. PMID: 11163683 Review.
-
Optical imaging of neuronal populations during decision-making.Science. 2005 Feb 11;307(5711):896-901. doi: 10.1126/science.1103736. Science. 2005. PMID: 15705844
-
Neural control of heartbeat in the leech and in some other invertebrates.Physiol Rev. 1979 Jan;59(1):101-36. doi: 10.1152/physrev.1979.59.1.101. Physiol Rev. 1979. PMID: 220645 Review.
Cited by
-
Sequential development of electrical and chemical synaptic connections generates a specific behavioral circuit in the leech.J Neurosci. 2005 Mar 9;25(10):2478-89. doi: 10.1523/JNEUROSCI.4787-04.2005. J Neurosci. 2005. PMID: 15758156 Free PMC article.
-
Imaging dedicated and multifunctional neural circuits generating distinct behaviors.J Neurosci. 2006 Oct 18;26(42):10925-33. doi: 10.1523/JNEUROSCI.3265-06.2006. J Neurosci. 2006. PMID: 17050731 Free PMC article.
-
A double-sided microscope to realize whole-ganglion imaging of membrane potential in the medicinal leech.Elife. 2017 Sep 25;6:e29839. doi: 10.7554/eLife.29839. Elife. 2017. PMID: 28944754 Free PMC article.
-
A hormone-activated central pattern generator for courtship.Curr Biol. 2010 Mar 23;20(6):487-95. doi: 10.1016/j.cub.2010.02.027. Epub 2010 Mar 11. Curr Biol. 2010. PMID: 20226670 Free PMC article.
-
Positive feedback loops sustain repeating bursts in neuronal circuits.J Biol Phys. 2011 Jun;37(3):317-45. doi: 10.1007/s10867-010-9210-8. Epub 2010 Dec 16. J Biol Phys. 2011. PMID: 22654180 Free PMC article.
References
-
- Berry MS, Pentreath VW ( 1976) Criteria for distinguishing between monosynaptic and polysynaptic transmission. Brain Res 105: 1-20. - PubMed
-
- Brodfuehrer PD, Burns A ( 1995) Neuronal factors influencing the decision to swim in the medicinal leech. Neurobiol Learn Mem 63: 192-199. - PubMed
-
- Brodfuehrer PD, Friesen WO ( 1986a) Initiation of swimming activity by trigger neurons in the leech subesophageal ganglion. I. Output connections of Tr1 and Tr2. J Comp Physiol [A] 159: 489-502. - PubMed
-
- Brodfuehrer PD, Friesen WO ( 1986b) Initiation of swimming activity by trigger neurons in the leech subesophageal ganglion. II. Role of segmental swim-initiating interneurons. J Comp Physiol [A] 159: 503-510. - PubMed
-
- Byrne JH, Castellucci VF, Kandel ER ( 1978) Contribution of individual mechanoreceptor sensory neurons to defensive gill-withdrawal reflex in Aplysia J Neurophysiol 41: 418-431. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
