Chemical chaperone therapy for brain pathology in G(M1)-gangliosidosis
- PMID: 14676316
- PMCID: PMC307667
- DOI: 10.1073/pnas.2536657100
Chemical chaperone therapy for brain pathology in G(M1)-gangliosidosis
Abstract
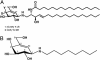
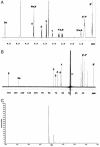
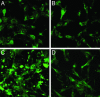
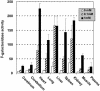
We synthesized a galactose derivative, N-octyl-4-epi-beta-valienamine (NOEV), for a molecular therapy (chemical chaperone therapy) of a human neurogenetic disease, beta-galactosidosis (GM1-gangliosidosis and Morquio B disease). It is a potent inhibitor of lysosomal beta-galactosidase in vitro. Addition of NOEV in the culture medium restored mutant enzyme activity in cultured human or murine fibroblasts at low intracellular concentrations, resulting in a marked decrease of intracellular substrate storage. Short-term oral administration of NOEV to a model mouse of juvenile GM1-gangliosidosis, expressing a mutant enzyme protein R201C, resulted in significant enhancement of the enzyme activity in the brain and other tissues. Immunohistochemical stain revealed a decrease in the amount of GM1 and GA1 in neuronal cells in the fronto-temporal cerebral cortex and brainstem. However, mass biochemical analysis did not show the substrate reduction observed histochemically in these limited areas in the brain probably because of the brief duration of this investigation. Chemical chaperone therapy may be useful for certain patients with beta-galactosidosis and potentially other lysosomal storage diseases with central nervous system involvement.
Figures

Similar articles
-
Chemical chaperone therapy: clinical effect in murine G(M1)-gangliosidosis.Ann Neurol. 2007 Dec;62(6):671-5. doi: 10.1002/ana.21284. Ann Neurol. 2007. PMID: 17994547
-
Fibroblast screening for chaperone therapy in beta-galactosidosis.Brain Dev. 2006 Sep;28(8):482-6. doi: 10.1016/j.braindev.2006.02.002. Epub 2006 Apr 17. Brain Dev. 2006. PMID: 16617000
-
Chemical chaperone therapy: chaperone effect on mutant enzyme and cellular pathophysiology in β-galactosidase deficiency.Hum Mutat. 2011 Jul;32(7):843-52. doi: 10.1002/humu.21516. Hum Mutat. 2011. PMID: 21520340
-
Chaperone therapy update: Fabry disease, GM1-gangliosidosis and Gaucher disease.Brain Dev. 2013 Jun;35(6):515-23. doi: 10.1016/j.braindev.2012.12.002. Epub 2013 Jan 3. Brain Dev. 2013. PMID: 23290321 Review.
-
Chemical chaperone therapy for GM1-gangliosidosis.Cell Mol Life Sci. 2008 Feb;65(3):351-3. doi: 10.1007/s00018-008-7470-2. Cell Mol Life Sci. 2008. PMID: 18202827 Free PMC article. Review.
Cited by
-
Chemical chaperones protect from effects of apoptosis-inducing mutation in carbonic anhydrase IV identified in retinitis pigmentosa 17.Proc Natl Acad Sci U S A. 2004 Aug 17;101(33):12300-5. doi: 10.1073/pnas.0404764101. Epub 2004 Aug 4. Proc Natl Acad Sci U S A. 2004. PMID: 15295099 Free PMC article.
-
Fabry disease - current treatment and new drug development.Curr Chem Genomics. 2010 Jul 23;4:50-6. doi: 10.2174/1875397301004010050. Curr Chem Genomics. 2010. PMID: 21127742 Free PMC article.
-
Lysosomal storage diseases: current therapies and future alternatives.J Mol Med (Berl). 2020 Jul;98(7):931-946. doi: 10.1007/s00109-020-01935-6. Epub 2020 Jun 11. J Mol Med (Berl). 2020. PMID: 32529345 Review.
-
Treating lysosomal storage diseases with pharmacological chaperones: from concept to clinics.EMBO Mol Med. 2009 Aug;1(5):268-79. doi: 10.1002/emmm.200900036. EMBO Mol Med. 2009. PMID: 20049730 Free PMC article. Review.
-
Sphingolipid lysosomal storage diseases: from bench to bedside.Lipids Health Dis. 2021 May 3;20(1):44. doi: 10.1186/s12944-021-01466-0. Lipids Health Dis. 2021. PMID: 33941173 Free PMC article. Review.
References
-
- Suzuki, Y., Oshima, A. & Nanba, E. (2001) in The Metabolic and Molecular Bases of Inherited Disease, eds. Scriver, C. R., Beaudet, A. L., Sly, W. S., Valle, D., Childs, B. & Vogelstein, B. (McGraw-Hill, New York), 8th Ed., pp. 3775-3809.
-
- O'Brien, J. S., Storb, R., Raff, R. F., Harding, J., Appelbaum, F., Morimoto, S., Kishimoto, Y., Graham, T., Ahern Rindell, A. & O'Brien, S. L. (1990) Clin. Genet. 38, 274-280. - PubMed
-
- Tylki-Szymanska, A., Maciejko, D., Kidawa, M., Jablonska-Budaj, U. & Czartoryska, B. (1985) J. Inherit. Metab. Dis. 8, 101-104. - PubMed
-
- Okumiya, T., Ishii, S., Takenaka, T., Kase, R., Kamei, S., Sakuraba, H. & Suzuki, Y. (1995) Biochem. Biophys. Res. Commun. 214, 1219-1224. - PubMed
-
- Fan, J. Q., Ishii, S., Asano, N. & Suzuki, Y. (1999) Nat. Med. 5, 112-115. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

