Safety evaluation of fixed combination piperaquine plus dihydroartemisinin (Artekin) in Cambodian children and adults with malaria
- PMID: 14678346
- PMCID: PMC1884411
- DOI: 10.1046/j.1365-2125.2003.01962.x
Safety evaluation of fixed combination piperaquine plus dihydroartemisinin (Artekin) in Cambodian children and adults with malaria
Abstract
Aims: To assess the haemodynamic, electrocardiographic and glycaemic effects of piperaquine-dihydroartemisinin (Artekin) fixed combination therapy in uncomplicated malaria.
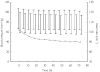
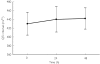
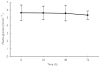
Methods: Sixty-two Cambodians (32 children and 30 adults) with falciparum or vivax malaria were given Artekin given as four age-based oral doses over 32 h. Supine and erect blood pressure, the electrocardiographic QT interval and plasma glucose were measured before treatment and then at regular intervals during a 4-day admission period as part of efficacy and safety monitoring. QT intervals were rate-corrected (QTc) using Bazett's formula.
Results: Artekin therapy was well tolerated and all patients responded to treatment. Average parasite and fever clearance times were 19 and 12 h, respectively. The pretreatment mean fall in systolic blood pressure on standing was 8 +/- 6 mmHg and 6-hourly measurements over 72 h showed no significant change (P = 0.48). There was a significant lengthening of the mean QTc to a maximum of 11 ms(0.5) (95% confidence interval 4-18 ms(0.5)) relative to baseline at 24 h (P = 0.003). The maximal QTc prolongation observed in any patient was 53 ms(0.5). There was a mean 0.4 mmol l(-1) reduction in the post-absorptive plasma glucose during the first 48 h but no episodes of hypoglycaemia (plasma glucose < 3.0 mmol l(-1)) were observed at any time.
Conclusions: Artekin is safe and effective combination therapy for uncomplicated malaria in children and adults. Although piperaquine is a long half-life drug related to other quinoline compounds including chloroquine and quinine, no clinically significant cardiovascular or metabolic effects were observed.
Figures
References
-
- White NJ. Delaying antimalarial drug resistance with combination chemotherapy. Parassitologia. 1999;41:301–308. - PubMed
-
- World Health Organisation. Antimalarial Drug Combination Therapy. Geneva: WHO/CDS/RBM; 2001. p. 35. Report of a WHO Technical Consultation.
-
- Denis MB, Davis TME, Hewitt S, et al. Efficacy and safety of dihydroartemisinin-piperaquine (Artekin) in Cambodian children and adults with uncomplicated falciparum malaria. Clin Infect Dis. 2002;35:1469–1476. - PubMed
-
- US Department of Health and Human Services. Guidance for industry. Food and Drug Administration; 1997. http://www.fda.gov/cder/guidance/index.htm.
-
- Zhao HJ, Xia YY, Zheng Z. Pre-clinical toxical study of new antimalarial agents. IV. Liver ultrastructure changes affected by antimalarial agent, compound tablet of piperaquine phosphate and sulfadoxine. Acad J Second Mil Med College. 1981;1:47–48.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

