Pathogenesis of flavivirus encephalitis
- PMID: 14689697
- PMCID: PMC7202458
- DOI: 10.1016/s0065-3527(03)60008-4
Pathogenesis of flavivirus encephalitis
Abstract
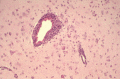
Within the flavivirus family, viruses that cause natural infections of the central nervous system (CNS) principally include members of the Japanese encephalitis virus (JEV) serogroup and the tick-borne encephalitis virus (TBEV) serocomplex. The pathogenesis of diseases involves complex interactions of viruses, which differ in neurovirulence potential, and a number of host factors, which govern susceptibility to infection and the capacity to mount effective antiviral immune responses both in the periphery and within the CNS. This chapter summarizes progress in the field of flavivirus neuropathogenesis. Mosquito-borne and tickborne viruses are considered together. Flavivirus neuropathogenesis involves both neuroinvasiveness (capacity to enter the CNS) and neurovirulence (replication within the CNS), both of which can be manipulated experimentally. Neuronal injury as a result of bystander effects may be a factor during flavivirus neuropathogenesis given that microglial activation and elaboration of inflammatory mediators, including IL-1β and TNF-α, occur in the CNS during these infections and may accompany the production of nitric oxide and peroxynitrite, which can cause neurotoxicity.
Figures







References
-
- Aihara H, Takasaki T, Toyosaki-Maeda T, Suzuki R, Okuno Y, Kurane I. T-cell activation and induction of antibodies and memory T cells by immunization with inactivated Japanese encephalitis vaccine. Viral Immunol. 2000;13:179–186. - PubMed
-
- Albrecht P. Pathogenesis of neurotropic arbovirus infections. Curr. Top. Microbiol. Immunol. 1968;43:44–91. - PubMed
-
- Aloisi F, Care A, Borsellino G, Gallo P, Rosa S, Bassani A, Cabibbo A, Testa U, Levi G, Peschle C. Production of hemolymphopoietic cytokines (IL-6, IL-8, colony-stimulating factors) by normal human astrocytes in response to IL-1 beta and tumor necrosis factor-alpha. J. Immunol. 1992;149:2358–2366. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
