Intrasteric control of AMPK via the gamma1 subunit AMP allosteric regulatory site
- PMID: 14691231
- PMCID: PMC2286513
- DOI: 10.1110/ps.03340004
Intrasteric control of AMPK via the gamma1 subunit AMP allosteric regulatory site
Abstract
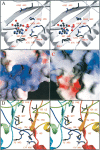
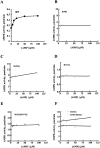
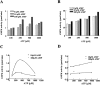
AMP-activated protein kinase (AMPK) is a alphabetagamma heterotrimer that is activated in response to both hormones and intracellular metabolic stress signals. AMPK is regulated by phosphorylation on the alpha subunit and by AMP allosteric control previously thought to be mediated by both alpha and gamma subunits. Here we present evidence that adjacent gamma subunit pairs of CBS repeat sequences (after Cystathionine Beta Synthase) form an AMP binding site related to, but distinct from the classical AMP binding site in phosphorylase, that can also bind ATP. The AMP binding site of the gamma(1) CBS1/CBS2 pair, modeled on the structures of the CBS sequences present in the inosine monophosphate dehydrogenase crystal structure, contains three arginine residues 70, 152, and 171 and His151. The yeast gamma homolog, snf4 contains a His151Gly substitution, and when this is introduced into gamma(1), AMP allosteric control is substantially lost and explains why the yeast snf1p/snf4p complex is insensitive to AMP. Arg70 in gamma(1) corresponds to the site of mutation in human gamma(2) and pig gamma(3) genes previously identified to cause an unusual cardiac phenotype and glycogen storage disease, respectively. Mutation of any of AMP binding site Arg residues to Gln substantially abolishes AMP allosteric control in expressed AMPK holoenzyme. The Arg/Gln mutations also suppress the previously described inhibitory properties of ATP and render the enzyme constitutively active. We propose that ATP acts as an intrasteric inhibitor by bridging the alpha and gamma subunits and that AMP functions to derepress AMPK activity.
Figures



References
-
- Adams, J.A. 2003. Activation loop phosphorylation and catalysis in protein kinases: Is there functional evidence for the autoinhibitor model? Biochemistry 42 601–607. - PubMed
-
- Barthel, A., Schmoll, D., Kruger, K.D., Roth, R.A., and Joost, H.G. 2002. Regulation of the forkhead transcription factor FKHR (FOXO1a) by glucose starvation and AICAR, an activator of AMP-activated protein kinase. Endocrinology 143 3183–3186. - PubMed
-
- Bateman, A. 1997. The structure of a domain common to archaebacteria and the homocystinuria disease protein. Trends Biochem. Sci. 22 12–13. - PubMed
-
- Blair, E., Redwood, C., Ashrafian, H., Oliveira, M., Broxholme, J., Kerr, B., Salmon, A., Ostman-Smith, I., and Watkins, H. 2001. Mutations in the γ(2) subunit of AMP-activated protein kinase cause familial hypertrophic cardiomyopathy: Evidence for the central role of energy compromise in disease pathogenesis. Hum. Mol. Genet. 10 1215–1220. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

