Linezolid pharmacokinetics in adult patients with cystic fibrosis
- PMID: 14693551
- PMCID: PMC310170
- DOI: 10.1128/AAC.48.1.281-284.2004
Linezolid pharmacokinetics in adult patients with cystic fibrosis
Abstract
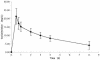
The pharmacokinetics of many drugs are altered in patients with cystic fibrosis (CF), often necessitating different dosage requirements than those used in non-CF patients. The objective of this study was to determine the pharmacokinetics of linezolid, an antibiotic with good activity against gram-positive organisms such as methicillin-resistant Staphylococcus aureus, in patients with CF so that dosage requirements could be established. Twelve adult patients (6 male) ranging in age from 22 to 39 years were studied. A single 600-mg dose was administered intravenously over 0.5 h, and plasma samples were collected at 0 (predose), 0.5, 0.75, 1, 2, 4, 8, and 24 h. Linezolid concentrations were determined with a validated high-performance liquid chromatography assay. Pharmacokinetic parameters were estimated using standard noncompartmental methods. Blood chemistry and hematologic indices were determined before and after the study for safety purposes. All patients completed the study without encountering any adverse reactions. The pharmacokinetic parameters, while variable, with half-lives varying from 1.76 to 8.36 h, were similar to those previously described in other populations. Mean (+/- standard deviation) values for pharmacokinetic parameters of interest were as follows: elimination rate constant, 0.21 (0.11) h(-1); half-life, 4.41 (2.43); volume of distribution at steady state, 0.87 (0.19) liters/kg of body weight; and total body clearance, 0.12 (0.06) liters/h/kg. No patient would have achieved the pharmacodynamic target of an area under the concentration-time curve/MIC ratio of 83 h for pathogens for which the MIC was 4 micro g/ml. Patients with inadequate clinical responses to linezolid may require more frequent dosing.
Figures
References
-
- Centers for Disease Control and Prevention. 2002. Staphylococcus aureus resistant to vancomycin—United States, 2002. Morb. Mortal. Wkly. Rep. 51:565-567. - PubMed
-
- Ferrin, M., J. B. Zuckerman, A. Meagher, and E. A. Blumberg. 2002. Successful treatment of methicillin-resistant Staphylococcus aureus pulmonary infection with linezolid in a patient with cystic fibrosis. Pediatr. Pulmonol. 33:221-223. - PubMed
-
- FitzSimmons, S. C. 1993. The changing epidemiology of cystic fibrosis. J. Pediatr. 122:1-9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical