Molecular and diagnostic clinical virology in real time
- PMID: 14706081
- PMCID: PMC7129573
- DOI: 10.1111/j.1469-0691.2004.00699.x
Molecular and diagnostic clinical virology in real time
Abstract
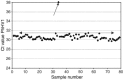
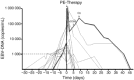
During the last decade, the application of both qualitative and quantitative nucleic acid detection techniques has had a major impact on diagnostics in clinical virology. Both signal and target amplification-based systems are currently used routinely in most if not all virology laboratories. However, commercial assays are only available for a very limited number of targets, and this has resulted in the development and introduction of assays developed in-house for most viral targets. With improved and automated nucleic acid sample isolation techniques, as well as real-time detection methods, a new generation of assays for most clinically important viruses is being developed. These technological improvements also make it possible to generate results with a very short turnaround time. As an example of a more individual-patient disease-management concept, we have introduced in our clinical setting the quantitative detection of Epstein-Barr virus (EBV) in T-cell-depleted allogeneic stem cell transplant patients. This has enabled us to develop models for pre-emptive anti-B-cell immunotherapy for EBV reactivation, and for reducing not only the incidence of EBV lymphoproliferative disease (EBV-LPD), but the virus-related mortality. It is now also feasible to introduce molecular testing for those viruses that can easily be detected using classical virological methods, such as culture techniques or antigen detection. Prospective studies are needed to evaluate the clinical importance of the additional positive samples detected. It should, however, be clear that a complete exchange of technology is unlikely to occur, and that complementary methods should stay operational, making possible the discovery of new viruses. Furthermore, the ability to characterise viruses more easily by sequencing opens new possibilities for epidemiological studies. There is also an urgent need, with regard to molecular diagnostic methods, for the introduction and use of standardised materials and participation in international quality control programmes. Finally, with the introduction of a universal internal control throughout the whole procedure, the accuracy of the results generated is warranted.
Figures
Comment in
-
Molecular and diagnostic clinical virology in real time.Clin Microbiol Infect. 2004 May;10(5):471; author reply 471-2. doi: 10.1111/j.1469-0691.2004.00905.x. Clin Microbiol Infect. 2004. PMID: 15113330 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

