Fyn and PTP-PEST-mediated regulation of Wiskott-Aldrich syndrome protein (WASp) tyrosine phosphorylation is required for coupling T cell antigen receptor engagement to WASp effector function and T cell activation
- PMID: 14707117
- PMCID: PMC1887720
- DOI: 10.1084/jem.20030976
Fyn and PTP-PEST-mediated regulation of Wiskott-Aldrich syndrome protein (WASp) tyrosine phosphorylation is required for coupling T cell antigen receptor engagement to WASp effector function and T cell activation
Abstract
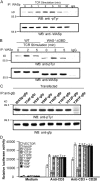
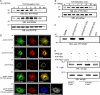
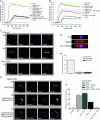
Involvement of the Wiskott-Aldrich syndrome protein (WASp) in promoting cell activation requires its release from autoinhibitory structural constraints and has been attributed to WASp association with activated cdc42. Here, however, we show that T cell development and T cell receptor (TCR)-induced proliferation and actin polymerization proceed normally in WASp-/- mice expressing a WASp transgene lacking the cdc42 binding domain. By contrast, mutation of tyrosine residue Y291, identified here as the major site of TCR-induced WASp tyrosine phosphorylation, abrogated induction of WASp tyrosine phosphorylation and its effector activities, including nuclear factor of activated T cell transcriptional activity, actin polymerization, and immunological synapse formation. TCR-induced WASp tyrosine phosphorylation was also disrupted in T cells lacking Fyn, a kinase shown here to bind, colocalize with, and phosphorylate WASp. By contrast, WASp was tyrosine dephosphorylated by protein tyrosine phosphatase (PTP)-PEST, a tyrosine phosphatase shown here to interact with WASp via proline, serine, threonine phosphatase interacting protein (PSTPIP)1 binding. Although Fyn enhanced WASp-mediated Arp2/3 activation and was required for synapse formation, PTP-PEST combined with PSTPIP1 inhibited WASp-driven actin polymerization and synapse formation. These observations identify key roles for Fyn and PTP-PEST in regulating WASp and imply that inducible WASp tyrosine phosphorylation can occur independently of cdc42 binding, but unlike the cdc42 interaction, is absolutely required for WASp contributions to T cell activation.
Figures



References
-
- Badour, K., J. Zhang, and K.A. Siminovitch. 2003. The Wiskott-Aldrich syndrome protein: forging a link between actin and cell activation. Immunol. Rev. 192:98–112. - PubMed
-
- Machesky, L.M., and R.H. Insall. 1998. Scar1 and the related Wiskott-Aldrich syndrome protein, WASP, regulate the actin cytoskeleton through the Arp2/3 complex. Curr. Biol. 8:1347–1356. - PubMed
-
- Rohatgi, R., L. Ma, H. Miki, M. Lopez, T. Kirchhausen, T. Takenawa, and M.W. Kirschner. 1999. The interaction between N-WASP and the Arp2/3 complex links Cdc42-dependent signals to actin assembly. Cell. 97:221–231. - PubMed
-
- Cory, G.O., R. Cramer, L. Blanchoin, and A.J. Ridley. 2003. Phosphorylation of the WASp-VCA domain increases its affinity for the Arp2/3 complex and enhances actin polymerization by WASp. Mol. Cell. 11:1229–1239. - PubMed
-
- Badour, K., J. Zhang, F. Shi, M.K. McGavin, V. Ramperasad, L.A. Hardy, D. Field, and K.A. Siminovitch. 2003. The Wiskott-Aldrich syndrome protein acts downstream of CD2 and the CD2AP and PSTPIP1 adaptors to promote formation of the immunological synapse. Immunity. 18:141–154. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

