Assessment of agreement between the AMPLICOR HIV-1 MONITOR test versions 1.0 and 1.5
- PMID: 14715766
- PMCID: PMC321658
- DOI: 10.1128/JCM.42.1.286-289.2004
Assessment of agreement between the AMPLICOR HIV-1 MONITOR test versions 1.0 and 1.5
Abstract
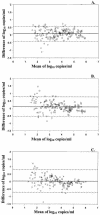
The agreement of the microwell plate AMPLICOR HIV-1 MONITOR version 1.0 (MWP 1.0), the microwell plate AMPLICOR HIV-1 MONITOR version 1.5 (MWP 1.5), and the COBAS AMPLICOR HIV-1 MONITOR version 1.5 (COBAS 1.5) tests was evaluated using clinical specimens and well-characterized control material. Two hundred patient plasma specimens and a panel of known human immunodeficiency virus type 1 (HIV-1) subtypes were tested. All data were log(10) transformed prior to analysis. The 95% limits of agreement for the three tests at the average of 3.66 log(10) copies/ml were +/- 0.28 log(10), +/- 0.34 log(10), and +/- 0.34 log(10) copies/ml for MWP 1.0-MWP 1.5, MWP 1.0-COBAS 1.5, and MWP 1.5-COBAS 1.5, respectively. Ten specimens (6.1%) had differences exceeding the limits of agreement for the MWP 1.0 and MWP 1.5 tests. Correlation coefficients among the three tests were high (r >or=0.96). The viral-load values obtained with the MWP 1.0 test were only 2.1% higher on average than those measured with the MWP 1.5 test and 1.6% higher than those seen with the COBAS 1.5 test. The MWP 1.5 test values were 0.8% higher than the COBAS 1.5 test values. Overall, there was less agreement among the different tests for viral-load values near the lower limit of quantification. The MWP 1.0 test underquantified subtypes A, E, F, G, and H by 1.0 to 2.0 log(10) copies/ml; this problem was not observed with the MWP 1.5 test. The close agreement among the results obtained with the different test versions and formats suggests that it is not necessary to reestablish a baseline viral load when changing AMPLICOR HIV-1 MONITOR tests, unless the patient is known to be infected with a non-B subtype.
Figures
References
-
- Bland, J. M., and D. G. Altman. 1986. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet i:307-310. - PubMed
-
- Brambilla, D., P. S. Reichelderfer, J. W. Bremer, D. E. Shapiro, R. C. Hershow, D. A. Katzenstein, S. M. Hammer, B. Jackson, A. C. Collier, R. S. Sperling, M. G. Fowler, and R. W. Coombs, for participating Adult and Pediatric AIDS Clinical Trials Groups, the Women Infant Transmission Study Clinics, and laboratories participating in the Virology Quality Assurance Program. 1999. The contribution of assay variation and biological variation to the total variability of plasma HIV-1 RNA measurements. AIDS 13:2269-2279. - PubMed
-
- Jagodzinski, L. L., D. L. Wiggins, J. L. McManis, S. Emery, J. Overbaugh, M. Robb, S. Bodrug, and N. L. Michael. 2000. Use of calibrated viral load standards for group M subtypes of human immunodeficiency virus type 1 to assess the performance of viral RNA quantitation tests. J. Clin. Microbiol. 38:1247-1249. - PMC - PubMed
-
- Lew, J., P. Reichelderfer, M. Fowler, J. Bremer, R. Carrol, S. Cassol, D. Chernoff, R. Coombs, M. Cronin, R. Dickover, S. Fiscus, S. Herman, B. Jackson, J. Kornegay, A. Kovacs, K. McIntosh, W. Meyer, N. Michael, L. Mofenson, J. Moye, T. Quinn, M. Robb, M. Vahey, B. Weiser, and T. Yeghiazarian, for the TUBE Meeting Workshop attendees. 1998. Determinations of levels of human immunodeficiency virus type 1 RNA in plasma: reassessment of parameters affecting assay outcome. J. Clin. Microbiol. 36:1471-1479. - PMC - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources